Review Article
1 Northern Arizona University, Flagstaff, Arizona, USA
2 Axolotl Biologix, Scottsdale, Arizona, USA
3 MedTech Incubator, Scottsdale, Arizona, USA
4 Arizona State University, Tempe, Arizona, USA
*Corresponding Author: Aaron J. Tabor, Medtech Incubator, Scottsdale, Arizona, USA.
Citation: David Cajthaml, Alison Ingraldi and Aaron Tabor, Amnion vs. Amnion/Chorion Placental Allografts: A Comparative Review of Composition, Mechanisms, and Clinical Use, J Clinical and Medical Research and Studies, V (5)I(2), DOI: 10.59468/2836-8525/154
Copyright: 2026 Aaron J. Tabor. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: April 24, 2026 | Accepted: April 28, 2026 | Published: April 30, 2026
Abstract
Placental-derived allografts are a regenerative technology often used as a protective barrier for tissues and a wound covering to promote wound healing in a variety of wound types. The allografts are derived from human placenta, which consists of the amnion and chorion layers. The innermost layer of the placenta, the amnion, natively functions as a strong protective barrier with anti-inflammatory, anti-bacterial, and angiogenic roles for the developing fetus. The chorion, which directly contacts and maintains communication at the fetal-maternal barrier, is rich in growth factors and cytokines which are implicated in angiogenic signaling, inflammatory regulation, and homeostasis. Placental-derived allografts are commonly formulated as either amnion-only or amnion and chorion containing allografts. Because these tissues have distinct native functions and compositions, understanding the underlying biology and mechanisms of each allograft is important for realizing their full clinical potential. Amnion-only allografts are generally thinner, and contain fewer angiogenic, inflammatory, and immunologic signaling factors. Amnion/chorion allografts contain more cytokines and growth factors, which can influence various healing mechanisms. Overall, both amnion and amnion/chorion allografts are a useful adjunct in treating various tissues and complex chronic wounds. Recognizing the differences and similarities between amnion and amnion/chorion allografts is crucial for providers to make informed decisions for their patients.
Keywords: Allograft; amnion; chorion; cytokine; inflammation; tissues; wound healing
Introduction
Wound care is a domain that is too often placed at the periphery of health care; however, with increasing prevalence and cost, it is quickly becoming a cornerstone of public health [1-3]. Estimating chronic wound prevalence and cost can be extremely challenging. Some estimates found that chronic wounds of mixed etiologies showed a global pooled prevalence of 2.21 per 1000 population [4]. With a world population of roughly 8.2 billion, this equates to about 18.12 million chronic wounds worldwide. In 2019, roughly one in six Medicare beneficiaries, about 10.5 million individuals, suffered from a chronic wound, which represents an increase in 2.3 million people since the previous update in 2014 [5]. In 2022, spending on wound care reached an estimated $148.65 billion in the United States alone, while China spent approximately $42.78 billion, and Japan roughly $22.91 billion on wound care [6]. Clearly, a shift in prioritization and advancements in chronic wound care is needed. An emerging approach to improve healing outcomes for chronic wounds is the use of placental tissue-derived allografts. These allografts serve as a valuable alternative in cases where chronic wounds exhibit slow or nonexistent healing progression, particularly for diabetic foot ulcers (DFUs) and venous leg ulcers (VLUs).
Placental membranes have been used clinically for decades as a biological matrix to support tissue repair. Their use has grown significantly in modern medicine due to advancements in procurement, processing, and preservation techniques. The first documented use of an amniotic membrane implantation dates back to 1910 (in skin grafting), with more border clinical adoption in the 1930s and 1940s where its implementation became more common (genital reconstruction and ophthalmology) [7]. Derived from human birth tissues, these grafts are typically available as either amnion-only or as combined amnion/chorion allografts. Across many indications, placental-derived allografts are valued for their ability to conform to tissue surfaces, provide a protective wound covering, and deliver biologically active factors that may influence inflammation, reepithelialization, angiogenesis, and matrix deposition and remodeling.
Currently, one of the most common applications for placental-derived allografts is in the management of chronic and acute wounds. Clinicians define a wound as chronic if it fails to progress in a predictable and timely manner within approximately 3 months [8]. The inability of chronic wounds to heal within an expected timeframe or progression complicates treatment and imposes a significant burden on patients. The complexity, increased morbidity, and cost associated with chronic wounds raise increasing concern and showcase the need for improved standard treatments. Clinically, the use of placental-derived membranes extends beyond chronic cutaneous ulcers to established and emerging applications, such as ocular reconstruction, burn and reconstructive surgery, oral and periodontal procedures, nerve and tendon associated repairs, and other indications where a protective and regenerative microenvironment is desired.
The placenta is composed of 2 layers: the amnion and chorion [9], [10]. Translationally, meaningful differences between amnion and amnion chorion allografts are plausible due to the tissue’s distinct native roles at the fetal-maternal interface. The amnion contributes tensile strength and barrier protection, whereas the chorion, being thicker and more cellular, participates more in immune and inflammatory signaling. However, little information directly comparing amnion and amnion/chorion allografts exist and it can be difficult to understand the differences and similarities of the two.
Despite widespread clinical use, direct head-to-head comparisons between amnion-only and amnion/chorion allografts remain limited, and clinicians often need to interpret assorted evidence across products that differ in processing, sterilization, preservation methods, compositions, and application protocols. Previous literature does not always disclose the exact composition and format of the placental-derived allografts used. In addition, limited comprehensive literature is available that describes the native roles and biological characteristics of the amnion and chorion. This makes it extremely challenging to separate tissue-level differences (amnion vs chorion) from product-level differences in manufacturing and their eventual clinical uses.
Accordingly, the goal of this manuscript is to provide a synthesis of existing literature to clarify the similarities and differences between amnion and amnion/chorion allografts. The intent is not to assert a general clinical superiority of one composition over the other but to deepen the understanding of their applications, mechanisms, and underlying fundamentals.
This review will first detail the anatomy and physiology of the amnion and chorion membranes, followed by an exploration of the biological activities relevant to each tissue. The paper will examine tissue sourcing, allografts, and the impact that processing has on biological activity. Subsequent sections will investigate direct comparisons of amnion and chorion in various fresh and processed tissue states. Next, published literature will be reviewed to identify the most common applications and outcomes associated with each allograft type. Finally, this review will provide comparative considerations, identify limitations and knowledge gaps, and propose future directions for more standardized, mechanism-linked evaluations.
2 - Placental Membrane Structure and Physiological Roles
The native structure and physiological characteristics of the tissues provide valuable insight into the distinctive roles of amnion and chorion within the human body. Understanding the functions and structures of these tissues is crucial, as these inherent characteristics may continue to influence the properties of processed allografts. Aristotle (384-322 BC) was among the first to recognize the amnion and chorion as essential fetal membranes, noting them as membranes that encase the fetus. However, it wasn’t until the 1870s, with advancements in microscopic observations that the origins of the amnion and chorion membranes were accurately identified [11]. The preceding sections will detail the anatomy and physiology of the amnion and chorion in their native roles.
2.1 – Anatomical Overview of the Human Placental Membranes
The human placental membranes, or fetal membranes, play a crucial role in the protection and nourishment of the fetus. These birth tissues are transient and exist only to support the fetus until birth [12], [13]. Collectively, these tissues provide mechanical protection against external shocks due to their collagen rich extracellular matrix (ECM). They also provide an immunological barrier for the fetus and participate in the regulation of amniotic fluid homeostasis [14]. There is an ongoing scientific debate regarding the number of distinct layers that compose these birth tissues. It is widely agreed upon that there are two primary layers: the fetal-facing amnion and the maternal-facing chorion. However, there is disagreement about whether the intermediate layer (spongy layer) between the two should be classified as a distinct and separate tissue [15-17]. Figure 1 illustrates the placental membranes enveloping the fetus (shown on the left) and provides a detailed breakdown of the amnion and chorion, along with their distinctive layers (shown on the right).
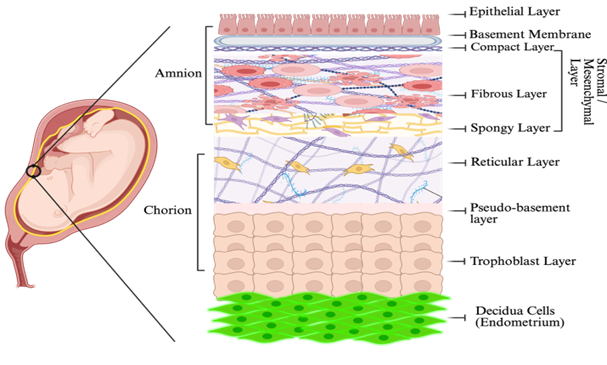
Figure 1. Illustration of the placental membranes within the uterus (left). Amnion and chorion schematic layer representation (right). Created with BioRender.com
Although universally recognized as a bilayer structure, the addition of the spongy layer (intermediate layer) separating the two is debated. Some authors consider it to be part of the amnion stromal compartment. These distinctions matter because each layer contributes differently to mechanical integrity, immune signaling, and extracellular matrix composition. These differences remain relevant when the tissues are processed into clinical allografts and considered for future medical applications.
2.2 - Amnion Native Structure and Physiological Role
The amnion membrane is the innermost layer of the placental membranes, directly contacting the amniotic fluid and encasing the developing fetus. This layer is composed of amniotic epithelial cells (AECs), a basement membrane, a compact layer, a fibrous layer containing amnion mesenchymal stem cells (MSCs), and a spongy intermediate layer [18]. Collectively, the compact, fibrous, and spongy layers are commonly referred to as the stromal layer. The amnion is a thin, semi-transparent tissue, often described as rough in texture, with thicknesses ranging from 0.02 to 0.5 mm [19], [20]. At full gestational development, the amnion layer has an average diameter of 18-22 cm, and the surface area of the placental disc measures between 200-400 cm2. However, the amniotic membrane extends beyond the placental disc. From conception to maturity, the amnion's appearance and structure vary. At term, the epithelial layer consists of a single layer of simple columnar cells directly over the placenta but transitions to a cuboidal shape in the extraplacental region [21]. Additionally, microvilli can be observed on the single layer of epithelial cells at term, though their presence is irregular and often appears branched [22]. Although these studies are primarily descriptive, the observed shift from columnar to cuboidal cells likely indicates some regional specialization of the amniotic epithelial layer, rather than a uniform cell population across the entire membrane.
The amnion serves multiple biologically active functions during pregnancy. It significantly contributes to the tensile strength of the fetal membrane, as its stroma is rich in collagen, which helps prevent stretching and premature rupture of the fetal membrane [16], [23]. Structurally, the amnion also helps contain the amniotic fluid, which cushions the fetus, prevents compression injuries, and allows for normal musculoskeletal development without the impediment of other structures [24]. Notably, the amnion is devoid of blood vessels and nerves, relying on diffusion from the chorion and the amniotic fluid to obtain nutrients and oxygen [25].
Despite its thin profile, the ECM of the amnion is composed of many biologically relevant proteins [26]. The amnion is rich in collagen types I, III, IV, V, VII, as well as laminin, nidogen, and fibronectin within its basement and compact layer membrane [27-29]. Importantly, the basement layer also contains hyaluronic acid and other critical ECM proteins that contribute key functional properties to the amnion, which will be discussed in future sections [18].
2.3 - Chorion Native Structure and Physiological Role
The chorionic membrane is located between the amnion and the maternal decidua/placental tissues. Like the amnion, the chorion is made up of distinctive layers. The reticular layer of the chorion, which contains chorionic mesenchymal stem cells, is in contact with the intermediate spongy layer of the amnion. Underneath this layer is a pseudo-basement layer, and finally the chorion trophoblast cells, which directly contact the maternal decidua cells. During most of the first trimester, the chorion is covered with villi, which ultimately regresses into a bilayer of trophoblasts [30]. During the early stages of pregnancy, the amnion and chorion are physically separated; however, the two tissues loosely fuse around 17 to 20 weeks of gestation [31].
While the chorion contributes to the barrier function of the placental membranes, it is primarily thought to serve as an immunological buffer, providing fetal defense against the maternal immune system [32]. Due to its direct contact with the maternal decidua, the chorion plays a crucial role in the fetal-maternal interface, maintaining immune tolerance and signaling pathways [33]. While the amnion is affected by both chemical and mechanical changes during gestation, the chorion remains largely unchanged, possibly preventing early degradation of the amnion during pregnancy [34].
In comparison, the chorion is thicker and more cellular than the amnion. Like the amnion, the chorion contributes to mechanical integrity under stretch and shear within the uterine environment. The chorion is more flexible and less stiff than the amnion, being reported to be twice as extensible, while possessing approximately ~60% of the strength of amnion [35]. The reticular layer forms much of the thickness for the chorion. The reticular network of fibers is primarily parallel, with nodes present at branching sites. Both fibroblasts and Hofbauer cells are present. The pseudo-basement layer forms the basement membrane for the trophoblasts and contains type IV collagen [16]. A dense fibrous connective tissue firmly adheres to the reticular layer, above which the trophoblasts can then anchor to. The outermost layer of the chorion consists of two to ten layers of trophoblast cells which contact the maternal decidua cells. While the amnion is typically described as avascular, the chorion is more closely associated with vascularized placental tissue [36].
The chorion’s reticular layer is rich in collagen types I, III, IV, V, and VI, as well as proteoglycans. The pseudo-basement layer contains collagen type IV, fibronectin, and laminin [37]. The chorion has been shown to be a rich source of 15-OH prostaglandin dehydrogenase (15-PGDH), which metabolizes prostaglandins and prevents their transmission from the amnion to the endometrium [38]. Reduced 15-PGDH activity can decrease prostaglandin metabolism, allowing them to pass to the decidua, where they persist and signal, potentially contributing to preterm birth stimuli [39].
2.4 - Native Summary Comparison
The amnion and chorion represent two structurally and functionally distinct components of the placental membrane. The amnion is the innermost layer facing the fetus, primarily acting as a thin barrier that provides support and contributes significantly to the tensile strength of the placental membranes. It is in direct contact with, and helps maintain, the protective amniotic fluid required for fetal development. The chorion, by comparison, is a thicker and more cellular layer positioned against the maternal decidua where it plays a role in immune buffering and signaling regulation, contributing to the overall strength and protection of the placenta. These differences indicate that each layer should be regarded as a specialized tissue with unique structural and physiological roles, which may influence how they behave as processed allografts.
3 - Biologically Relevant Activities of Amnion and Chorion Membranes
The amnion and chorion have been investigated for various biomechanisms that support their native roles and are likely relevant to their downstream implementation as processed allografts. Both tissues exhibit inhibitory/excitatory pathways and produce many biologically active factors that give these tissues inflammatory, immune, angiogenic, and homeostatic influences. Acknowledging these distinctive properties of the amnion and chorion is crucial as they may continue to influence such pathways during their implantation as an allograft.
3.1 - Biologically Relevant Activities of the Amnion
Anti-Inflammatory, anti-scarring, and immunomodulatory functions
The amnion is distinguished by its potent anti-inflammatory and immunomodulatory activity. It suppresses excessive inflammation through the production and secretion of anti-inflammatory cytokines, such as IL-10, and through the expression of key inflammatory pathway inhibitors, including IL-1 receptor antagonist (IL-1RA) and protease inhibitors (e.g., α1-antitrypsin and inter-α-trypsin inhibitor) [42]. Amniotic epithelial cells have been reported to release immunomodulatory mediators including PGE2, indoleamine 2,3-dioxygenase (IDO), and hepatocyte growth factor (HGF), which contribute to local immune regulation [43]. In addition to biochemical signaling, the amnion acts as a physical barrier capable of trapping infiltrating inflammatory cells and inducing apoptosis within its matrix, thereby supporting the healing process and resolution of inflammation [44].
Hyaluronic acid (HA), and hydrated proteoglycans are abundant throughout the amnion [45], [46]. High molecular weight HA extracted from the amnion can covalently bind with the heavy chains (HC) of inter-α-inhibitor (IaI), forming HA-HC complexes that suppress TGF-β1 promoter activation and induce macrophage apoptosis. These mechanisms together likely contribute to the amnion's well-documented anti-inflammatory and anti-scarring effects [47], [48]. The amniotic basement membrane also contains perlecan, a multifunctional proteoglycan that stabilizes the ECM through interaction with fibronectin, laminin, and many collagens, while playing a role in cell adhesion as well [49]. Fibrosis, a pathological outcome of chronic inflammation, is characterized by excessive ECM component buildup driven largely by dysregulated TGF-ß signaling [50]. The amnion naturally suppresses myofibroblast overactivation, excessive collagen deposits, and fibrotic remodeling [51]. In a 2016 rat model of biliary fibrosis, after 6 weeks, amnion-treated animals exhibited significantly lower fibrosis than the control rats, with collagen deposition, activated myofibroblasts and TGF-β1 all reduced to approximately 50% the level in untreated rats [52]. These effects are attributed to modulation of TGF-ß signaling and the unique ECM composition of the amnion [53-55].
Molecular analyses have found that the amnion expresses TGF-β3, a cytokine associated with regenerative, low-scarring healing [56], [57]. While TGF-β1 and TGF-β2 promote collagen deposition, increasing scar formations, a higher TGF-β3:TGFβ-1 ratio helps promote regenerative healing over heavy ECM deposition [58]. Overall, the amnion downregulates TGF-ß pathway components including TGF-β1, TGF-β2, TGF-β3, and TGF-Receptor II thereby limiting excessive ECM production [59]. These regulatory mechanisms help prevent chronic inflammation and fibrosis during pregnancy and likely contribute to the amnion's therapeutic effects when used as an allograft tissue [60]. TGF-β signaling is key to fibroblast proliferation and subsequent ECM production and thus the inhibition of TGF-β limits excessive fibroblast activity and aids in reducing scarring and ECM buildup [59]. The anti-scarring properties of the amnion are further supported by its secretion of tissue inhibitors of metalloproteinases (TIMPs) and its ability to maintain a balanced matrix metalloproteinase (MMP) environment, promoting organized tissue regeneration [19]. The amnion also limits fibroblast differentiation into myofibroblasts, which are often considered a key driver for fibrotic tissue formation [59].
Beyond its immunomodulatory and antifibrotic functions, the amnion exhibits broad antibacterial and antimicrobial properties. It contains bactericidin, ß-lysin, lysozyme, transferrin, and 7S immunoglobulin, all of which provide microbial protection [61], [62]. The presence of these soluble factors inhibit pathogens commonly found in chronic wounds, including P. aeruginosa, S. aureus and methicillin-resistant S. aureus (MRSA) [63]. Amniotic epithelial cells also produce β-defensins, secretory leukocyte protease inhibitors (SLPI), and elafin antimicrobial peptides with additional immunoregulatory roles [24]. Lactoferrin, present in the amnion tissue, provides further antimicrobial and anti-inflammatory protection, serving as an antioxidant [64]. A 2016 clinical study of 33 burn patients treated with amnion allografts reported no significant wound infections, which may support the membrane’s protective antimicrobial effects [65].
Angiogenic Properties
Angiogenesis is the formation of new blood vessels from existing vasculature and is essential for embryonic development, tissue repair, and physiological remodeling of capillaries [66]. Angiogenesis can also contribute to pathological processes such as tumor progression, where expansion of the vascular network is crucial for metastasis and increased metabolic demands [67]. Studies of the amnion reveal interesting properties in relation to angiogenesis. The epithelial side of the amnion demonstrates anti-angiogenic effects, with the amniotic epithelial cells producing several inhibitory angiogenic-related factors, including endostatin, TIMP-1 through TIMP-4, and thrombospondin [26], [68], [69]. These cells also produce IL-1 receptor antagonists, which indirectly suppress IL-1-mediate pro-angiogenic signaling [66].
Simultaneously, the stromal layer of the amnion supports angiogenesis. Amniotic mesenchymal stem cells secrete vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), IL-8, angiogenin, interferon-γ, IL-6, basic fibroblast growth factor (bFGF), and epidermal growth factor (EGF), all of which contribute to neovascularization and modulate wound healing [56], [60], [70]. Amniotic mesenchymal cells can also promote angiogenesis via differentiation into endothelial cells [27].
A 2013 rat study investigated the side-dependent effects of amnion allograft transplantation, where a layer of dorsal skin was removed, and an amnion allograft was applied either epithelial side down (n=8) or stromal side down (n=8). When applied epithelial side down, vessel sprouting decreased relative to controls; when applied stromal side down, vessel sprouting increased. In an additional aortic ring assay, angiogenic activity of fibroblast cells was only observed after removal of the amniotic epithelial cells, regardless of graft orientation [71]. These findings highlight the functional duality of the amnion and underscore the importance of graft orientation and cellular retention when clinical goals involve either promoting or limiting neovascularization.
3.2 - Biologically Relevant Activities of the Chorion
Immune Homeostasis, Pro-inflammatory Role, and Angiogenesis
The chorion plays a pivotal role in maintaining immune homeostasis at the maternal-fetal interface. The chorion trophoblasts secrete human chorionic gonadotropin (hCG), promoting the induction and activation of regulatory T cells (Tregs), a critical mechanism for establishing maternal immune tolerance during pregnancy. hCG also suppresses the differentiation of Treg cells into proinflammatory Th17 cells, a lineage associated with fetal rejection and impaired growth [72]. Furthermore, hCG also inhibits CXCL10 expression by influencing H3K27me3 methylation which subsequently binds the CXCL10 promoter, suppressing its expression. CXCL10 is a potent chemoattractant for cytotoxic CD8+ T cells and is elevated during infection and preterm birth; therefore, its suppression contributes to fetal protection [73].
Chorion trophoblasts further support immunomodulation by producing anti-inflammatory hormones, cytokines, and by buffering decidual immune cell migration [16]. Additionally, the expression of human leukocyte antigen G (HLA-G) within the chorion contributes to maternal-fetal immune tolerance by inhibiting NK cell and T-cell cytotoxicity [74].
Despite these immunoregulatory functions, the chorion also plays an active role in the pro-inflammatory cascade during gestation. Compared with the amnion, the chorion produces more pro-inflammatory cytokines. Chorionic trophoblasts secrete IL-1β and IL-18 during the second trimester through inflammasome activation [75]. Inflammasome signaling plays a critical role in host defense against numerous pathogens, including Listeria monocytogenes, a bacterium capable of crossing the maternal-fetal interface and causing fetal infection and, in severe cases, premature death [76].
Chorion cells also mount a robust antiviral response. When exposed to the influenza virus, chorion cells upregulate IL-1β, IL-6, TNF-α, IFN-β, IFN-γ and GM-CSF in response to the infection, while this effect is not observed in amnion cells. Infected chorionic cells are subsequently cleared through apoptosis-dependent phagocytosis by recruited macrophages. This coordinated interaction between cytokine-secreting chorion cells and maternal monocytes/macrophages within the decidua implies the chorion forms an important defensive barrier against viral invasion. Growing evidence indicates that fetal chorion cells serve a central role in protecting the maternal–fetal interface from influenza infection by promoting pro-inflammatory cytokine expression and programmed cell death. During the early stages of viral replication, chorion cells initiate apoptotic pathways that eliminate infected cells, thereby limiting viral propagation. Concurrently, these cells release factors that promote monocyte differentiation [77]. This pro-inflammatory response is increasingly recognized as a key mechanism by which the chorion protects the maternal-fetal interface from viral pathogens.
The pro-inflammatory environment generated by the chorion also contributes to physiological processes required for parturition. During the early stages of pregnancy, a pro-inflammatory environment is required for the implantation of the blastocyte. This environment is also required for trophoblast invasion into the decidua, since the trophoblasts must traverse the endometrial ECM, blood supply, and adjacent tissues [78].
Pro-inflammatory cytokines including IL-1α, IL-1β, IL-6, and TNF-α stimulate the production of prostaglandin E2 (PGE2) and matrix metalloproteinases (MMPs), which promote uterine contractions and degrade collagen within the fetal membranes. IL-6 widens intercellular gaps in amniotic epithelial cells, contributing to membrane weakening, while TNF-α, particularly when combined with IFN-γ, induces apoptosis in placental and fetal membrane cells. IL-1β and TNF-α further activate pro-apoptotic pathways involving p53 and caspase-dependent apoptotic pathways. Increased apoptosis, especially during chorioamnionitis, can weaken the structural stability of the membranes, and these cytokines have been repeatedly implicated in triggering preterm labor, underscoring their central role in the initiation of parturition [77].
In addition to its immunological functions, the chorion exhibits notable pro-angiogenic activity. Compared with the amnion, the chorion contains higher levels of pro-angiogenic cytokines [79]. Chorionic trophoblasts express angiogenin and VEGF, both contribute to vascularization and fetal development [80]. These angiogenic signals contribute to placental vascularization and may influence tissue repair responses when chorion-derived allografts are used clinically.
3.3 - Key Biological Contrasts Between Amnion and Chorion
The amnion and chorion are not two interchangeable tissues; rather, they serve distinct and complementary biological roles within the fetal membranes. Although they function together as a bilayer system during gestation, their distinctive structural composition, cellular activity, and biochemical signaling and biomechanisms show the importance of analyzing them separately for their translation into allografts and their applications.
The amnion is commonly described as a protective, anti-inflammatory, and regulatory matrix that supports epithelial integrity and helps limit fibrosis, whereas the chorion is generally thicker and more cellular, functioning as an immunologically active interface that contributes to signaling at the fetal–maternal boundary and participates in immune signaling and angiogenic pathways.
Table 1. Summary of relevant key characteristics for the amnion and chorion with sources.
| Amnion Key Characteristic | Sources | Chorion Key Characteristics | Sources |
| Dual Angiogenic Properties (epithelial anti-angiogenic; stromal pro-angiogenic) | 26, 27, 56, 60, 66, 68, 69, 70, 71 | Predominantly pro-angiogenic; high VEGF and angiogenin expression | 79, 80 |
| Immune Regulation; Anti-bacterial/antimicrobial peptide & protein production | 24, 60, 61, 62, 63, 64, 65 | Important immunoregulatory role at the fetal-maternal interface (hCG, HLA-G, Treg induction) | 16, 72, 73, 74 |
| Anti-Inflammatory factor production; anti-fibrotic ECM composition | 40, 41, 42, 45, 46, 47, 52, 53, 54, 55, 56, 57, 58, 59 | Pro-inflammatory cytokine production; inflammasome activation; pathogen defense | 75, 76, 77, 78 |
| Anti-Scarring effects via TGF-β regulation, HA-HC complexes, & reduced myofibroblast activation | 19, 48, 59 | Higher cytokine and growth factor content; supports trophoblast invasion and placental development | 101, 102, 103 |
4 - Transformation of Placental Membranes into Allografts
Once the native structure, function, and biological characteristics of amnion and chorion are understood, it becomes essential to examine how these tissues are processed into clinically usable allografts. Placental-derived allograft processing varies among manufacturers, but most follow the same general procedure. The desired final product composition, amnion-only, chorion-only, multilayer constructs, or combined amnion/chorion membranes also influences the processing techniques used. These variations complicate direct comparisons between products, but understanding the native biology of each membrane provides a foundation for interpreting how processing may alter their behavior and clinical performance. Understanding the native composition of the amnion and chorion is important as this knowledge can be used to infer and make scientific justification on how a specific allograft composition and processing technique may influence its behavior and efficacy in real-world clinical applications.
4.1 - Tissue Sourcing and Processing
The preparation of placental-derived allografts is crucial as subtle changes can impact downstream functions and efficacy of the final products. Allografts are grafts harvested and transplanted between genetically non-identical individuals of the same species [81]. For humans, common allografts include products such as tendons, cortical bone struts, demineralized bone matrices, and amniotic membranes. Allografts are often minimally processed and are frequently transplanted to serve their original innate function. These tissues are often used by surgeons and clinicians with over 1 million implants of allografts annually [82]. Human placental-derived allografts most often function as wound coverings to serve as a barrier, like the placental role as a protective barrier to the developing fetus.
To make placental-derived allografts, donated tissue must first be collected. Consenting donors must first complete comprehensive medical and serological screening. Health screening protocols vary among tissue collecting banks, but overall, they cover the same precautions. The placenta is collected from consenting mothers who chose to donate to tissue banks. Typically, these are from cesarean sections, as these allow the tissue to be obtained using aseptic techniques. However, an increase in vaginal collections is occurring within the donor field. Donors are screened for required diseases such as human immunodeficiency virus (HIV), syphilis, hepatitis B and C, and other transmissible diseases [83]. A complete health history review and serologic panel is required to ensure safety and minimize the transmission of infectious diseases by licensed medical providers.
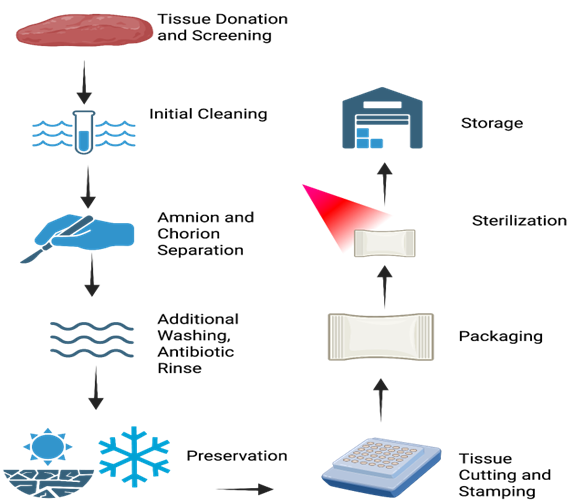
Figure 2. Generalized workflow for placental-derived allograft manufacturing, illustrating the major processing steps from tissue recovery, cleaning, separation, preservation, and sterilization. Created using BioRender.com
After collection, the tissue undergoes a series of standardized steps performed under aseptic conditions to prevent contamination. Processing steps vary by manufacturer, but they all follow the same general processing flow. The tissues are first washed to remove any residual blood, clots, or residual debris [83]. Depending on the manufacturing needs, the amnion and chorion are separated and then the tissues can be discarded, left intact, or folded to achieve the desired product composition (single/dual layer, amnion-only, chorion-only, amnion/chorion, etc.). The tissue goes through an additional final washing cycle before being preserved and cut to the desired allograft size. The cutting procedure may include stamping to denote membrane orientation. Preservation methods include cryopreservation, hydration, air dried dehydration/oven dehydration, or lyophilization [84-86]. Once cut to size, the processed allografts are packaged and sterilized. Sterilization is commonly done through gamma irradiation, but additional techniques include supercritical CO2 and chemical sterilization. The sterilized allografts are then approved and stored before allograft disbursement to end clinicians.
Despite the importance of these steps, many published studies omit key processing details, including layer composition, decellularization status, preservation method, and sterilization technique. This lack of transparency complicates interpretation and comparison across products. These details are important to be able to interpret and apply results to other products that may or may not be similar.
4.2 - Impact of Processing on Biological Activity
One major point of variation among placental-derived allografts is whether the tissue is used in a decellularized or non-decellularized form. Decellularization is a process used to remove the cellular components from the tissue while preserving the ECM structure and various cytokines and growth factors. This process creates a non-immunogenic natural scaffold that may reduce transplant rejection, increase allograft integration, and support host cell migration [87-90].
Decellularization can be achieved through chemical, enzymatic, or physical methods [87], [88]. While removal of cellular material may reduce foreign-body reactions, it can also alter the ECM architecture and diminish native bioactivity and composition of allografts. This is one of the main reasons why it is important to find a technique that balances the removal of cellular components while maintaining the desired biological activity and structure integrity. This processing technique may reduce immunogenic cellular material, but it may also diminish retained growth factors, proteoglycans, and the process can add a significant time and cost burden, potentially altering the regulatory status of the allograft [91], [92]. Due to these considerations, some manufacturers intentionally choose to produce products that remain non-decellularized to best preserve the native bioactivity and structure integrity, while reducing costs and manufacturing complexity.
Differences in preservation, decellularization, and sterilization methods can significantly alter the biochemical composition, structural integrity, and functional properties of placental-derived allografts. These changes affect not only the composition of the allografts themselves, but also its clinical application and performance. A 2010 study conducted by Lim et al. compared decellularized dehydrated human amniotic membrane (DDHAM) to cryopreserved human amniotic membrane (CHAM) comparing the chemical makeup, histology, and clinical efficacy of the two [93]. The DDHAM exhibited basement membrane disruption and loss of collagen IV and VII, laminin, and fibronectin when compared to the CHAM. The DDHAM also showed lower levels of epidermal growth factor receptor, fibroblast growth factor 2, insulin like growth factor I, platelet-derived growth factor A, platelet-derived growth factor B, transforming growth factor (TGF) 1, TGF-β, TGF-2 receptor, and vascular endothelial growth factor. Despite these biochemical differences, upon clinical application of the DDHAM to an ocular surface, rapid epithelialization was observed, and the treatment was deemed successful; demonstrating that structural and biochemical alterations do not necessarily preclude clinical efficacy.
In one of the final processing stages of allograft manufacturing, the product must be sterilized. Multiple sterilization techniques exist for placental-derived allografts including the most common technique, gamma irradiation. Additional sterilization techniques include supercritical CO2 and various chemical sterilization techniques such as peracetic acid/ethanol treatment [94]. Gamma irradiation has a long-standing history of sterilization in the medical world and was first approved in 1963 by British Pharmacopoeia [95]. Gamma irradiation has been shown to disrupt epithelial layers, damage fibroblast structures, and reduce growth factor levels, such as TGF-β and bFGF [96-97]. Newer techniques such as supercritical carbon dioxide (scCO2) look to offer an alternative to gamma irradiation. A 2025 study developed a scCO2 sterilization technique that offered similar sterilization results as gamma irradiation but better maintained the tissues’ structure and various growth factors [98].
A 2020 proteomics analysis conducted by Becktell et al. analyzed 9 different amniotic membrane and amniotic-derived products from four different manufacturers [99]. Although total protein counts were similar between two amniotic membrane allografts (470 vs 473 proteins), the composition of proteins was vastly different and they only shared five of the ten most abundant proteins. This highlights how even in products that appear identical and are advertised the same, differences in manufacturing, donor biological variability and processing can change the composition of allografts.
While the goal of this review was not to compare the effect of processing methods on the biological activity of placental-derived membranes, these cases and existing literature suggest that processing has an effect. It has been demonstrated that processing alters the proteomic and physical structure of placental-derived allografts. Therefore, it is important to consider the processing technique used with placental-derived allografts, as there are significant differences between the various techniques. Further research is needed to identify the best processing methods. Despite variations in composition and structure, multiple processing techniques are effective and have been reported to improve healing outcomes in many studies.
4.3 - Why Processing Can Complicate Direct Comparisons
The heterogeneity of processing methods – and the inconsistent reporting of these methods – makes direct comparison of placental-derived allografts challenging. Even at the level of raw tissue, amnion and chorion can be referred to in conjunction as amniotic tissues or amniotic membranes, without specifying whether the authors are referring to amnion, chorion, or both layers, unless explicitly stated. This ambiguity extends into the allograft literature, where products may be described simply as “amniotic membrane allografts” without clarifying which tissue membrane(s) they contain, making the comparisons of amnion and chorion quite difficult.
Even when layer composition of the placental-derived allografts is stated, both for clinical applications and in vitro analysis, important details on the composition or processing of an allograft are often left out. Even in cases when an allograft is explicitly described as amnion, amnion-chorion, or a specific combination of layers, there is still some degree of vagueness in details such as hydration status, preservation method, decellularization, washing procedures, sterilization technique and storage condition. Formulations exist of dual/multi-layer amnion, and amnion/chorion allografts, and these are intermittently referred to in the same way as a single-layer formulation. All of these may affect the efficacy of an allograft during clinical studies and applications, or the biochemical and structural properties for research. Without a complete technical description, comparisons between products, even those marketed similarly, become difficult, complicating the comparisons between amnion-only and amnion/chorion allografts.
5 - Comparative Evidence Across Tissue States
Now that an understanding of processing techniques and their variation has been established, it is appropriate to discuss direct comparisons of amnion and chorion. The literature on the topic is limited and often difficult to interpret, as discussed above. Because of this, comparisons will include both raw unprocessed amnion and chorion, processed amnion and chorion, and amnion and amnion/chorion allografts specifically. In doing so, the native differences between the amnion and chorion will be highlighted and can be seen persisting through processing to influence the characteristics of each allograft.
5.1 - Growth Factor and Cytokine Profile Differences
Wound healing is a complex, multi-staged process which is influenced by various cytokines and growth factors [100]. The characterization of these factors within the amnion and chorion helps to enhance the understanding of the type of impact these allografts may have on various mechanisms throughout the body. Across different comparisons, amnion/chorion preparations tend to show higher signaling content, largely because the chorion is thicker and carries a higher signaling load [101-103]. In an evaluation of 18 growth factors and cytokines, amnion/chorion allografts possessed a 4-5-fold higher growth factor content compared to amnion-only allografts [101]. In this direct comparison of processed amnion and chorion allografts which included TIMP-1, TIMP-2, TIMP-4, bFGF, PIGF, EGF, IL-4, IL-6, and others, both membrane types had detectable amounts of each, but the amnion-only allograft only contained more PIGF (118.72 pg/mg and 114.63 pg/mg respectively) and EGF (77.08 pg/mg and 5.13 pg/mg respectively) compared to the amnion/chorion allograft. Another comparison of fresh amnion and chorion found that fresh amnion had significantly higher levels of GAL-7, TGF-β1, and IL-1F5 while chorion had significantly higher levels of EG-VEGF, PDGF-BB, and TIMP-2 [102]. These differences suggest that chorion containing allografts may provide a denser signaling load, which may have implications for clinical implementation.
5.2 - Structural Factors
The structural composition and distribution of ECM components of amnion and amnion/chorion allografts can give insight into their biological activity. A 2022 study investigated dehydrated and sterilized amnion (dsAM) and amnion/chorion (dsAC), focusing on the distribution of structural factors, including nuclei, elastin, collagen I, collagen III, polysaccharides, glycosaminoglycans (GAG’s), fibronectin, and laminin [104]. GAGs were evenly distributed through the basement membrane of the dsAM, and between the two basement membranes and the trophoblast layer of the dsAC. Collagen distribution was seen through the dsAM with minimal elastic fiber distribution. Within the dsAC, collagen is evenly distributed and elastin is seen primarily within the basement membrane of the chorion. Fibronectin was visualized extensively in both the dsAM and the dsAC. These findings are important because the organization of ECM components may influence the grafts characteristics. Clinically, differences may help explain why amnion and amnion/chorion allografts may demonstrate distinct performance profiles across wound types.
Collagen I was strongly expressed in the dsAM below its basement membrane and well distributed within the intermediate layer of the dsAC, with minimal expression in the chorionic trophoblast layer. Collagen III was faintly visualized in the dsAM intermediate layer and along connective tissue tendrils within the dsAC. This distinction is relevant because collagen I contributes primarily to ECM strength and stability, whereas collagen III is more closely associated with tissue remodel and repair. These differences in collagen distribution may influence graft handling characteristics as well as the regenerative environment for each allograft type.
Laminin was lightly expressed in the dsAM but well visualized throughout the dsAC with visualization through the compact layer of the amnion. In this study, the authors hypothesized that processing and separation of the amnion and chorion had large effects on laminin content based on the differences and location of laminin expression. This is particularly important because laminin plays a central role in cell adhesion and migration. Therefore, differences in laminin preservation during processing may have a meaningful impact on epithelial support, tissue integration, and graft performance.
5.3 - Inflammation and Immunomodulation Related Signaling
Inflammation and immune-modulating signaling also plays a role in the wound healing cascade, controlled by an intricate balance of cytokines and growth factors [105], showcasing the importance of characterizing the differences across allograft compositions. Analyses comparing amnion and chorion suggest that the inflammatory signaling profile depends not only on membrane type, but also on whether the tissue is evaluated in a fresh unprocessed state or after preservation and processing. An analysis of immune-modulating growth factors and cytokines (IL-1F5, IL-1RA, IL-4, IL-6, IL-8, IL-10, TIMP-1, TIMP-2, TIMP-3, TIMP-4, and TNF-α) found that overall, unprocessed fresh chorion contained slightly higher levels of such factors than unprocessed fresh amnion per mg [103]. Fresh amnion did show higher levels of IL-1F5, IL-6, and IL-8 [103]. However, a similar study of processed amnion and chorion found higher levels of IL-6 and IL-8 in chorion tissue. The differences could once again be associated with differences in processing or donor variability [101]. Taken together, these findings suggest that while chorion may generally carry a greater total inflammatory and immune-signaling load, specific cytokine retention may shift depending on how the tissue is processed.
Fresh and unprocessed chorion showed higher levels of IL-1RA, IL-4, IL-10, TIMP-1, and TIMP-2. This broader trend supports the idea that amnion/chorion allografts may deliver a larger inflammatory and immune-signaling payload than amnion-only allografts. Amnion-only allografts may be optimal when a lighter biological load is preferred, while amnion/chorion allografts may be favored when a more robust signal-heavy environment is desired. These differences may be relevant when selecting an allograft for wounds or surgical environments where either a more restrained or a more biologically active signaling profile is preferred.
5.4 - Protease Regulators
Matrix metalloproteinases (MMPs) are often present in excess in chronic wounds. Excessive or poor regulation of MMPs can lead to excessive ECM degradation faster than deposition can occur resulting in non-healing chronic wounds [106]. Tissue inhibitors of metalloproteinases (TIMPs) modulate ECM remodeling and MMP activity, and the ratio of TIMP to MMP influences wound healing [107]. Across both fresh and processed comparisons, chorion generally appears to retain a greater protease-regulating burden than amnion, although the magnitude of this difference varies by tissue state and study.
Processed amnion and chorion tissues show similar levels of TIMP-4 (5999.2 and 5958.0 pg/mg), while processed chorion has significantly more TIMP-1 (4.49 and 18.77 pg/mg) and TIMP-2 (227.8 and 378.0 pg/mg) [101]. Similarly, unprocessed fresh amnion has been shown to have similar levels of TIMP-4, with decreases in TIMP-1 and TIMP-2 [102]. However, both dehydrated and frozen amnion have been shown to contain significantly less TIMP-1, TIMP-2, TIMP-3, and TIMP-4 than their chorion counterparts in an additional study [103]. This again highlights that different states of tissue processing techniques may influence protein composition. Tissue-to-tissue or biologic variation may also play a role in the heterogeneity of the results.
Overall, it appears that amnion/chorion membranes would deliver a greater mass of protease inhibitors per cm2. From an application standpoint, this could make chorion-containing grafts more attractive in protease-rich wound environments where excessive ECM breakdown is a concern. Additional research should be conducted on MMP inhibition in vivo to investigate the effects of TIMP presence in amnion and amnion/chorion allografts on protease regulation.
5.5 - Angiogenesis
Investigation into angiogenic signaling molecule differences in amnion and chorion tissues yielded mixed findings, although the overall trend again appears to differ according to whether tissues are fresh or processed. Following similar trends, the unprocessed fresh chorion contains higher pro-angiogenic signaling molecules when compared to unprocessed fresh amnion [102]. A panel of growth factors and cytokines including VEGF, PDGF, and bFGF, were labeled as
“angiogenic” and pg/mg levels were measured for each tissue type. In all cases, the chorion showed higher levels of these angiogenic signaling molecules, leading to an overall heightened pro-angiogenic signaling profile [102]. In an earlier mentioned proteomic comparison, VEGF and IL-8 (commonly considered pro-angiogenic factors) were found to be significantly higher in processed chorion vs amnion [101].
Controversially, when looking at EGF levels, fresh or processed amnion levels are significantly higher than their chorion counterparts [101-103]. EGF may indirectly support angiogenesis but is not generally considered pro-angiogenic like VEGF. This distinction is important because it suggests that chorion could provide a stronger overall vascular signaling environment, while amnion may retain select factors that support repair through somewhat different mechanisms. Given these results and the generalization that chorion-containing allografts contain higher angiogenic factors than amnion-only allografts, amnion/chorion combinations may be preferred when granulation and vascular support are top priorities. Amnion-only might be preferred when a thinner, lower-total-load membrane is preferred with less angiogenic signaling density.
Table 2. Generalized differences in the signaling profile between amnion and chorion, fresh and processed placental tissues.
| Fresh Amnion | Fresh Chorion | Processed Amnion | Processed Chorion | |
| Inflammation / Immunomodulation | Lower overall burden; select IL-1F5, IL-6, and IL-8 higher | Slightly higher overall immune-signaling load | Mixed / processing-dependent | Often higher retained IL-6 and IL-8; greater total load |
| Protease Regulators (TIMPs) | Lower TIMP-1 and TIMP-2; TIMP-4 similar in some studies | Higher overall TIMP burden than fresh amnion | Lower than processed chorion in multiple studies | Higher TIMP-1 and TIMP-2; TIMP-4 similar in some comparisons |
| Angiogenic Signaling | Lower overall proangiogenic profile; EGF often higher | Higher overall proangiogenic signaling profile | Lower VEGF / IL-8 than processed chorion; EGF may remain higher | Higher VEGF / IL-8 and greater overall angiogenic signal density |
5.6 - Thickness and Usability
Direct comparative data on the thickness for amnion and amnion/chorion allografts are limited. Processing techniques (dehydration, lyophilized, cryopreservation, sterilization, etc.) all influence the thickness of processed allografts as well as the specific layer composition. However, natively the amnion has a reported thickness of 0.02 to 0.5 mm [19], [20] while the chorion has been reported to be approximately 3-4 times thicker than the amnion [104].
Given that the chorion is thicker, this means that amnion/chorion allografts are generally thicker than amnion-only allografts. Commercially processed amnion/chorion composite allografts (especially multi-layer constructs) can be hundreds of microns thick, and a tri-layer amnion/chorion configuration has been reported to be ~3x thicker than a bilayer configuration [108]. The thickness differences are important considerations with some calling the amnion thickness an advantage for its “easy handling, lightweight, elasticity, and adhesiveness" [109] while others point to “problematic handling and suturing” [110]. It stands to reason that amnion-only may be preferred when maximum conformability and minimal bulk are desired, while amnion/chorion configurations may be preferred for more substantial wrap-like handling situations.
5.7 - Translation into Clinical Considerations
While direct comparisons of placental-derived tissues and allografts are limited, it is still possible to draw conclusions for clinical applications based on repeated outcomes across tissue states and products. These conclusions can be applied to clinical application relevance and offer some considerations of amnion and amnion/chorion allografts based on repeated themes across the literature, summarized in figure 3.
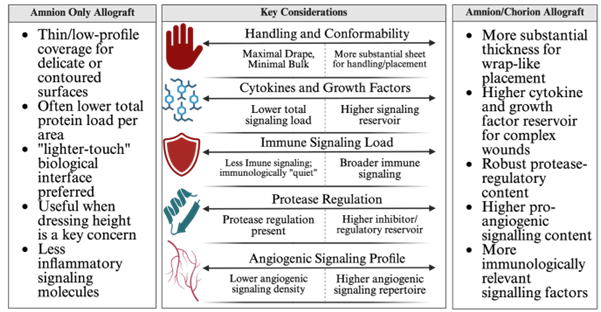
Figure 3. Clinician selection considerations for amnion-only and amnion/chorion allografts, Created in https://BioRender.com. The figure presents some possible considerations for clinicians to consider when deciding between an amnion-only or amnion/chorion allograft.
Several considerations for the clinical application of amnion and amnion/chorion allografts are present including conformability, cytokine/growth factor load, and signaling profile. With the presented differences and possible considerations for amnion and amnion/chorion allografts, it is important to investigate the current literature surrounding the use of each allograft type. This will aid in highlighting where the membrane allografts are currently being used in clinical practice, and where these allografts appear to be having the most success.
6 - Reported Use of Amnion or Amnion/Chorion Allografts in the Literature
6.1 - DFUs and VLUs
When investigating the clinical application of amnion or amnion/chorion allografts for efficacy in treating chronic wounds, significant research focuses on diabetic foot ulcers (DFUs) and venous leg ulcers (VLUs), for a multitude of reasons. First, DFUs and VLUs are among the most common chronic wounds. In a real-world epidemiology analysis of lower extremity ulcers (which affect 49 million people worldwide), DFUs and VLUs were the most common types encountered in clinical practice [111]. DFUs alone affects millions of individuals annually with a prevalence of around 19-34% in those with diabetes [112]. VLUs affect roughly 1.69% of the elderly population, and their economic burden in the U.S. is estimated to be around $14.9 billion annually [113]. Because of their relatively high prevalence, DFUs and VLUs have a well-understood and distinctive pathophysiology. DFUs arise through metabolic dysfunction, impaired immune response, vascular insufficiency, and neuropathy [114], [115]. VLUs on the other hand are driven by chronic venous insufficiency and hypertension which leads to edema and inflammation [116]. As DFUs and VLUs have a well-understood pathophysiology, their treatment is also quite standardized, positioning them as an extremely useful model for research. They have standard inclusion criteria, reproducible endpoints, and can be easily compared against standard care [117]. As such, many investigations in the efficacy of amnion and amnion/chorion allografts on the treatment of chronic wounds use DFUs and VLUs as a reliable and reproducible model.
6.2 - DFU and VLU Amnio-Derived Allograft Applications
The following studies are not presented as a comprehensive review of all available evidence but were selected because they represent recent clinical experience across multiple study designs. Together, they illustrate how amnion-only allografts are being applied alongside the standard of care in the treatment of DFUs and VLUs. The presented studies were selected for their robust design and more recent findings. Additional trials and systematic reviews report comparable outcomes and further detail [118-122].
- A 2021 randomized control pilot trial investigated the use of a dried human amniotic membrane (dHAM) allograft for its effectiveness as an addition to standard care for the treatment of DFUs. Thirty-one patients (mean age 59.8 years,87% with type 2 diabetes) were randomized to receive either standard care with placebo dHAM or standard of care plus dHAM applied epithelial side up twice weekly. Outcomes were assessed at 12 weeks or upon wound closure. Within 12 weeks, 4 (27%) of index ulcers in the dHAM group healed versus 1 (6.3%) in the control group. Percentage wound area reduction was also greater in the dHAM group, supporting the potential benefit of dHAM as an adjunctive therapy. [123].
- A 2014 case study from the Mercy Hospital Outpatient Wound Care and Hyperbaric Medicine Center evaluated the effectiveness of a dehydrated amniotic membrane allograft (DAMA) on treating non-progressing chronic wounds. This case study enrolled 7 patients (mean age 64, 57?male) who presented with VLUs (n=4), surgical (n=2), or DFU (n=1) chronic wounds. Patient inclusion criteria indicated the wounds had to be non-progressing with a minimum of 4 weeks of unsatisfactory progress utilizing standard care practices. The preferred method of DAMA application was an initial sharp debridement and confirmation that the wounds were free from clinical signs of infection. DAMA was trimmed and applied to the wound margins with ~1mm of overlap and wrapped in dressing following the standard of care for each wound type. All wounds had received prior care including debridement with topical collagenase, NPWT (negative pressure wound therapy), offloading and topical antibiotics, and applications of another CTP (cellular or tissue-based product) with multiple rounds of HBOT (hyperbaric oxygen therapy). DAMA applicationwas weekly to biweekly based on clinical evaluation with an average of 8 days between applications. After a single application, 86% of patient wounds demonstrated size reduction. Overall, 87% of patients achieved wound closure, with 43% closing after only three DAMA applications. Mean time to closure was 7.9 weeks, suggesting DAMA may be effective in uncooperative chronic wounds [124].
- A prospective clinical trial published in 2023 investigated the efficacy of a viable human amnion membrane allograft (vHAMA) on treating non-healing DFUs. During the trial, patients underwent a 2-week screening process during which they received standard of care (SOC) treatment. At the end of the 2 weeks, those whose wounds did not improve by 20% were included in the study. A total of 20 patients were enrolled following the 2-week screening period. Wounds were treated with a weekly application of vHAMA in addition to SOC for up to 12 weeks. During weekly visits the wounds received sharp debridement, were cleaned, and analyzed for the study. Of the 20 patients, 10 healed by week 6 and 17 healed by week 12. For the 3 patients that did not heal, two were withdrawn for adverse events not related to the use of vHAMA. The mean time to heal was 46.6 days and an average of 5.4 vHAMA applications were received [125].
6.3 - DFU and VLU Amnion/Chorion-Derived Allograft Application
Similarly, the studies outlined below do not represent a comprehensive review of the available literature. Rather, they highlight recent applications of amnion/chorion allografts in the treatment of DFUs and VLUs with emphasis on larger RCTs when available. Additional studies and systematic reviews provide broader context and product-specific details beyond the scope of this section [126-130].
- A randomized control trial was conducted from 2014 to 2017 as a prospective analysis on the effectiveness of dehydrated human amnion/chorion membrane (dHACM) allografts on the treatment of DFUs. A total of 218 patients were enrolled in the study across 14 United States centers. All patients had DFUs below the ankle and were first subjected to a run-in period. During the run-in period patients were taught standard care practices and were evaluated weekly for progress. At the end of the 2 weeks, those with wounds that had a reduction in size of less than 25% moved on to the 12-week treatment phase. After run-in period exclusions and additional exclusion criteria, 110 patients were randomly divided into the treatment (n=54) and control group (n=56). The treatment group received weekly dHACM allografts in addition to standard care and offloading while the control group received standard care and offloading. At the end of the 12 weeks, 70% of the dHACM-treated ulcers had completely healed compared to 50% of the no-dHACM group. Healing rates at 12 weeks were 81% for the dHACM group and 55% for the no-dHACM group, for those who finished the trial. Furthermore, of the 38 healed wounds in the dHACM group, 95% remained closed at the 16-week follow-up. Of the 28 healed wounds in the no-dHACM group, 86% remained closed at the 16-week follow-up [131].
- A randomized controlled study was conducted from March 2015 to March 2017 to investigate the efficacy of a dehydrated amnion/chorion membrane (dHACM) allograft on the treatment of VLUs. A total of 189 subjects were screened and entered a 2-week run-in period. At the end of the trial, 109 subjects were ultimately included for analysis which included 52 in the dHACM group and 57 in the standard of care control group. Subjects in the dHACM group received up to 12 weekly applications of the allograft in addition to a standard moist wound dressing and multilayer compression bandage. Subjects in the standard of care group received a moist wound dressing and a multilayer compression bandage. All subjects had weekly visits, cleaning and debridement for 12 weeks of the treatment phase and presented for a follow-up visit at week 16. At the conclusion of the 12-week treatment phase, 60% of subjects in the dHACM group had completely healed VLUs compared to 35% in the standard care group. Mean percentage wound area reduction was 66% in the dHACM group compared to 40% in the control group at the end of the 12 weeks. At the 16-week mark, mean VLU area reduction was 72% in the dHACM group and 39% in the control group [132].
- A 2024 randomized controlled trial investigated the efficacy of a dehydrated amnion chorion membrane (dACM) on treating hard-to-heal DFUs. An initial 438 subjects were enrolled, however after inclusion/exclusion screening was complete 218 subjects moved forward. 109 subjects were randomly assigned to the dACM plus standard of care (SoC) group and 109 subjects were assigned to the SoC group. Those in the dACM group received the dACM directly on the ulcer bed chorion side down at weekly intervals in addition to SoC which included sharp debridement and sterile saline cleanings. The control SoC group received the same SoC but not dACM. Subjects in both groups were seen weekly for up to 12 weeks. The dACM treated group had a median complete wound closure (CWC) time of 84 days. The control group did not achieve a median time to CWC during the 12 weeks. dACM treatment increased the probability of CWC by 48% compared to the control SoC-only group. The mean percent reduction in wound area from baseline was 72% for the dACM group and 21% for the SoC-only group at the end of the 12 weeks [133].
6.4 - Further Clinical Applications for Amnion-Derived Allografts
Beyond DFUs and VLUs, amnion-derived allografts have a long clinical history and a rapidly expanding evidence landscape across multiple clinical specialties. Modern literature supports their use as a bioactive and comfortable membrane that can promote epithelialization, modulate inflammation, with adoptions seen in ophthalmology, reconstructive surgery, oral/maxillofacial procedures, and musculoskeletal and nerve related applications [134-137]. The following studies showcase the broad clinical use of amnion-derived allografts.
- A prospective randomized clinical trial was completed between February 2009 and October 2009 to investigate the efficacy of a processed amnion allograft on the treatment of split-thickness skin graft patients who underwent burn wound reconstructive surgery. A total of 42 patients with second- or third-degree burn wounds with a total body surface area (TBSA) of 20-40% were enrolled in the trial and randomly assigned to the amnion treatment group (n=21) and the control group (n=21). Each patient underwent a standard split-thickness skin graft to cover the burn wounds. Those in the amnion group received the membrane as a wound covering following standard wound bed preparation and then covered with a sterile gauze over the membrane. The control group received the same wound bed preparation, and the wound was covered with a conventional Vaseline-containing dressing. Wounds were assessed daily, and control group subjects had daily bandage changes. The amnion group frequency of changing and reapplication was not explicitly reported. Analysis was completed after 4 weeks of treatment. The duration of wound healing was 17.61 days in the amnion group compared to 21.16 days in the control group, which represented statistical significance. Wound healing was defined as reepithelialization of the wound, confirmed by spontaneous detachment of the wound dressing or scaling of the amniotic membrane. Patient cooperation score was 1.6 in the amnion group and 2.93 in the control group, which again was significant. The authors suggest amnion membrane as an alternative to standard of care as it provided significant benefits in increasing patient comfort by changing the number of dressing changes and accelerating wound healing [138].
- A randomized controlled trial was conducted to investigate the effects of an amnion allograft intervention after free gingival graft surgery. The study enrolled 27 patients of which 15 were randomized in the amnion group and 12 were randomized into a control group. The amnion membrane group received amniotic membrane dressing sutured over the palatal donor site while the control group did not receive anything over the suture site. Analysis was conducted at 7, 14, and 21 days post-operatively and was completed by 3 periodontists. Pain was assessed via a visual analog scale (VAS) and analgesics were noted. Amoxicillin, anti-inflammatories, and a 0.2% chlorhexidine digluconate mouthwash were prescribed to each patient. The mean color match scores were higher in the test group than the control at 14 and 21 days (both p-values <0>
- A case study published in 2016 evaluated the use of an amniotic membrane transplantation (AMT) for fornix reconstruction to improve ocular prosthesis retention. The case study evaluated 18 patients, all of which had blind eyes with obliterated fornices and were unable to retain a prosthesis. All patients had fornix reconstruction surgery using AMT. During the surgery, subconjunctival scar tissue was removed along with the removal of cysts and cicatricial tissue when necessary. A layer of HAM was applied epithelial side face up over the exposed bare episclera. An antibiotic-steroid drop ointment was prescribed for 2 weeks, and lubrication continued for up to 4 months. Complete success was defined as the restoration of an adequate deep fornix with the retention of the prosthesis. Partial success was deemed as the focal recurrence of scar and symblepharon with the prosthesis still retained. Complete success was seen in 15 patients with 1 case of partial success and 2 cases of failure [140].
- A randomized clinical trial conducted between September 2014 and November 2015 investigated the efficacy of a human amniotic membrane (HAM) on its effects for wound healing in patients after an anal fistulotomy. A total of 73 patients were enrolled presenting with a clinical diagnosis of a low type fistula. The 73 patients were randomly assigned to the HAM group (n=37) and the control group (HAM). All patients received the same fistulotomy and curettage procedure. Those in the HAM group had the HAM applied and fixed on the side of the wound in four points the same as marsupialization. Those in the control group received standard marsupialization done in four points of the side wall. Wounds in both groups then had a surgical dressing applied. The dressing was removed after day 1 and patients were discharged on day 2 following they had no complications. The patients were visited 3, 7, 14, 21, and 28 days post-operative. A visual analog scale (VAS) was used to assess itching, gas, stool incontinence, discharge, and pain. Photographs were taken intraoperative, and again on the second visit. Tissue biopsies were taken from 10 patients randomly selected in each group. At 14 days post-operative the mean ± SD of percent recovery was 67.39 ± 4.69 in the HAM group and 54.51 ± 4.86 in the control group. The difference was statistically significant. No differences in pathology were observed. Fecal incontinence increase was statistically significant in the HAM group compared to the control group. Discharge, pain, and itching, was lower in the HAM group [141].
6.5 - Further Clinical Applications for Amnion/Chorion-Derived Allografts
Amnion/chorion derived allografts have been applied across a widening range of acute and reconstructive indications, burns, fistulas, and ocular surface defects. Recent literature suggests an expansion into dermatology and oral/maxillofacial surgery [142-144]. The use cases for amnion/chorion derived allografts appear similar to amnion-only allografts, even with the early described differences. The studies below are not intended to be exhaustive, rather they provide representative examples of use cases beyond DFUs and VLUs.
- A case study conducted between January 1st, 2016, and June 30th, 2023, enrolled 16 patients with inflammatory bowel disease (IBD). The authors investigated the efficacy of a dehydrated human amnion chorion membrane (DHACM) allograft on preventing colorectal anastomotic leaks (AL) by use as a protective barrier. All 16 patients received the DHACM applied directly on a primary colorectal anastomosis without a protective ostomy. The endpoint of the study was a 90-day post operative re-evaluation for incidences of AL (formally defined as reanastomosis, stent, colostomy, drainage, fluid collection, or abscess). Within this cohort of 16 patients, there were no reported incidents of AL, rehospitalizations, adverse side-effects or deaths. A comparison to the Premier Healthcare Database and National Surgical Quality Improvement Program showed AL leak rates for similar procedures without the use of DHACM at 6.2% and 6.0% respectively [145].
- A case control study published in 2018 investigated the effects of a dehydrated human amnion chorion membrane (dHACM) augmented anterior cruciate ligament (ACL) allograft used for ligament reconstruction. A total of 15 patients underwent ACL reconstruction with tripled peroneus augmented dHACM and were compared with 15 control patients who did not receive the dHACM augmentation. Post-operative analysis was conducted at two, four, six, and eight months post-op. Tenor-Lysholm scores were assessed at each check in and a 6-month post-operative MRI signal-to-noise (SNR) was taken by mean signal value at femoral, midsubstance, and tibial insertion of the ACL grafts. A single surgeon utilized the same surgical technique for all ACL reconstruction following standard protocol. The dHACM allograft was lightly rehydrated with saline and wrapped around the ACL graft as a sleeve and stitched in place. The mean time for graft curing in the dHACM group was 5 months, compared to 8.6 months in the control group. Initial post-operative mean Lysholm scores were 46.3 for the dHACM group and 45 for the control group (not statistically significant). At eight months post-operative average Lysholm scores for the dHACM group were 92.3 and 83.3 (significant difference). The SNR from the MRI at 6 months showed 9 subjects in the good healing category (SNR <75> 150) for the dHACM group. In the control group 5 patients were in good healing, 6 were in moderate healing, and 4 were in poor healing [146].
- A case series conducted between August 2015 and August 2016 investigated the use of a micronized dehydrated human amnion chorion (dHACM) allograft as an injectable to treat pain associated with tendinopathy or arthritis. A total of 50 patients presented with a clinical diagnosis of degenerative joint/tendon pathology and a well-documented clinical record of pain and function status prior to the treatment. After exclusions due to declining interview follow-up and contact issues, 40 patients received the treatment. All 40 patients received 40mg of micronized dHACM suspended in 1mL of saline solution. 20 patients received a joint injection, and 20 patients received a tendon injection. After injection patients were told to avoid strenuous activity and the use of NSAIDs. Data was collected on worst and average reported pain before the procedure and at 1, 2, and 3 months post-procedure. No adverse side effects were reported however, local pain was present in 72.5% of patients with a mean of 2.25 days of pain post-procedure. Average reported baseline pain was 6.4 (95% CI = 5.7 - 7) and decreased to 2.7 at 1 month (95% CI = 2.1 - 3.3), 1.7 at 2 months (95% CI = 1.1 - 2.2), and 1.4 at 3 months (95% CI = 0.9 - 1.9). A clinically meaningful improvement in pain was defined as a 30% reduction in average pain. At 1 month, 75% of patients achieved this, 84% at 2 months, and 91% in 3 months. Finally, patient reported functional impairment was a 6.8 (on the ADL subscale of the PSFS) (95% CI = 6 - 7.5) before the procedure. At the 3-month follow-up the mean functional impairment score decreased to a low level of 2 (95% CI = 1.4 - 2.7) [147].
- A 2015 study published in 2015 evaluated the use of a dehydrated human amnion chorion allograft membrane (dHACM) and its effects on soft tissue scarring in the epidural space in patients having transforaminal lumbar interbody lumbar fusion (TLIF). Patients received standard TLIF with posterior pedicle screw fixation along with a dHACM cut to size and placed in the epidural space. A total of 5 patients received the dHACM treatment. After adequate fusion was achieved, the patient underwent instrument removal with an exploration of the epidermal space. During the exploration, assessment of scar formation was completed by the surgeon using a 4-level scoring system evaluation adhesion to tissue (1 = no adhesion, 4 = sharp dissection required). A 1cm2 section of lumbar tissue was removed adjacent to the epidural space for histological assessment of scar tissue formation. During tissue removal 4 out of 5 patients had an adhesion score of 2 (easily detachable) while 1 patient had an adhesion score of 4. Angiolipoma of 5 was noted in 2cases and 10% was noted in 1 case while 0% was noted in the other 3 cases. Patient reported outcomes had significant improvements and no intraoperative or post-operative complications were reported. The authors note the small sample size but suggest that dHACM implantation during TLIF may reduce epidural fibrosis and is generally well tolerated [148].
6.6 - Evidence for Efficacy Overview
The studies and cases summarized in Sections 5.2-5.6 are not intended to be comprehensive but rather they provide a representative overview of where amnion and amnion/chorion allografts are currently being applied and how they are commonly evaluated in the literature. Across both allograft preparations, DFUs and VLUs remain the most heavily studied indications and provide the most standardized framework for evaluating their efficacy in treating chronic wounds. These wounds have well established pathology and standard of care regimens with common endpoints and quantitative measurement to easily compare studies. Consequently, DFU and VLU trials, particularly large RCTs when available, provide the most comparable evidence for interpreting clinical evidence.
Outside lower extremity ulcers, clinical use spans multiple specialties including ocular reconstruction, burn and reconstructive surgery, oral/maxillofacial procedures, gastrointestinal and colorectal application, spine procedures, and musculoskeletal contexts as a barrier, injectable or tissue wrap. These reports support the view that placental-derived allografts are being used as a broadly applicable biologically active membrane.
Importantly, the clinical literature evaluates amnion and amnion/chorion as categories even though product performance is influenced by preservation/processing techniques, graft format, dosing, orientation, and composition. These factors create difficulty in evaluating the measured outcomes, making it hard to generalize results across studies and tissue types.
Interestingly, the use cases across both allograft types appear quite similar. As described earlier, the native origin, physiological properties, and biomolecular composition of amnion and chorion have differences that don’t seem to be explicitly utilized throughout their use in clinical applications. It might be assumed given their differences there could possibly be noticeable clinical differences between these allograft formulations, but they don’t appear to be fully explored at this point. Without adequate head-to-head comparisons of both in vitro and in vivo analysis it is difficult for physicians to separate the clinical efficacy of various placental-derived allograft formulations. This highlights the need for more robust comparisons of the mechanistic properties and biological behaviors of amnion and amnion/chorion allografts.
7 - Comparative Considerations
7.1 - Shared Biological Features
Despite the aforementioned structural and compositional differences between amnion and amnion/chorion allografts, both constructs share several core features that likely set the foundation for their broad and often similar utility across the mentioned applications. Both tissues and subsequent allografts are ECM rich biological membranes that natively function as protective barriers and can provide the same supportive and protective characteristics as allograft applications. They both provide a supportive mixture of cytokines, growth factors, and protease-regulating factors that can influence cell migration and tissue remodeling. Notably however, even “same-label” membrane allografts can still have different proteomic composition which reinforces the idea that biological activity reflects both the source tissue and processing choices.
7.2 - Distinct Structural and Signaling Characteristics
A repeated theme across the literature in direct comparisons of allograft compositions is that amnion/chorion preparations tend to carry a higher total signaling load, plausibly driven by the increase in thickness given by the chorion. This increase in cytokines and growth factors may include higher levels of inflammatory/immune modulators, matrix remodeling factors, and pro-angiogenic signaling. Amnion-only allografts are generally thinner and have a lower total-load of signaling biomolecules which could be advantageous when conformability and minimal bulk are high considerations.
Angiogenesis-related signaling represents a nuanced example across the measure differences within the literature. Evidence summarized earlier showed that chorion (and thus chorion-containing preparations) can show higher levels of pro-angiogenic factors like VEGF and IL-8. This trend is not uniform however with different literature suggesting that amnion may contain higher levels of EGF compared to chorion. Conclusively, the angiogenic potential should not be treated as a single axis metric, but rather that the membrane choice could influence the type and balance of angiogenic signals.
From a practical standpoint, thickness and handling are also important considerations. Natively different thicknesses and processing dependent alterations can possibly lead to a meaningful variation in the clinical pliability, suturability, conformability, and wrap-like behavior.
7.3 - Implications for Biological Function and Uses
Because chronic wounds vary in inflammatory burden, vascular insufficiencies, and protease composition it is important to consider amnion vs amnion/chorion not as a binary “best option” but rather viewed on a spectrum that should be evaluated. In this spectrum-like viewpoint,Amnion-only allografts may be favored in scenarios where a thin, highly conformable barrier with a lighter overall signaling load density with minimal bulk is preferred. Amnion/chorion allografts may be preferred when a higher overall total growth factor/cytokine load with more robust handling characteristics are of higher clinical priority.
8 - Limitations and Knowledge Gaps
Several limitations constrain the direct comparison of amnion and amnion/chorion allografts and complicate interpretation of the presented data, and available evidence. First, substantial manufacturing heterogeneity exists across products. Differences in dehydration versus cryopreservation, decellularization, and sterilization methods can alter the physical structure, growth factor, and cytokine composition of the final graft product. These variations make cross-study comparisons difficult and limit the ability to apply generalized findings, even among products labeled similarly.
Second, relatively few research studies are designed as true head-to-head comparisons in which amnion and amnion/chorion allografts are evaluated under matched preparation, processing, and clinical conditions. As a result, most comparisons across the literature are indirect, drawing from studies with different patient populations, methodologies, and outcome measurements. Third, biologically active signaling content measurement does not necessarily translate directly to bioavailability at the treatment site, and many factors play a role in the effect these allografts have on the healing process.
Finally, many mechanistic hypotheses proposed throughout the literature - including roles in inflammatory regulation, protease modulation, angiogenic influence, require more robust in vivo validation. Even when total protein content appears similar across products, the abundance of individual proteins can vary substantially including donor-to-donor variability. This underscores the need for more standardized characterization and reporting when interpreting amnion and amnion/chorion allografts as broad categories.
9 - Conclusions and Future Directions
Placental membrane allografts represent a clinically valuable class of biomaterials with a broad range of applications already being explored in the chronic wound space and other soft-tissue indications. The literature demonstrates that both amnion and amnion/chorion allografts function effectively as protective wound barriers and provide biologically active compounds that support tissue repair, epithelization, and modulation of the wound environment. Differences in thickness/handling and the overall “signaling load” between the two membrane types are plausibly linked to the chorion’s native structure and processing-dependent alteration introduced during allograft manufacturing. Looking ahead, several research directions would meaningfully strengthen the evidence base surrounding amnion and amnion/chorion allografts to improve clinical decision-making:
- Standardized head-to-head comparisons of amnion and amnion/chorion allografts utilizing identical processing techniques in both in vivo and in vitro settings.
- Mechanism-linked outcome studies connecting the measured differences in inflammatory, angiogenic, and protease-balance signaling to wound bed biomarkers and clinically relevant healing outcomes.
- Bioavailability and kinetics analyses to clarify how compositional differences translate into in vivo signaling, including release profiles, degradation behavior, and how compositional differences translate into wound signaling in vivo.
- Improved reporting standards in clinical studies, including detailed product characterization and specifications, graft size and dosing frequencies, and quantitative/qualitative endpoint alignment for easier cross-study comparisons.
Overall, the current evidence does not support a binary “best-choice”, between amnion-only and amnion/chorion allografts. Instead, a more practical and considerate view, these products occupy different positions within a range of thickness, handling, signaling density, and signaling composition. Optimal allograft selection should be guided by the biological needs and mechanistic constraints of the wound environment or intended tissue application, recognizing that manufacturing and processing techniques can drive variability and contribute to noticeable differences in performance. Continued mechanistic and comparative research will be essential for refining these distinctions and enhancing clinical precision in allograft selection.
References
- Falanga, V., Isseroff, R. R., Soulika, A. M., Romanelli, M., Margolis, D., Kapp, S., Granick, M., & Harding, K. (2022). Chronic wounds. Nature reviews. Disease primers, 8(1), 50.
- Sen C. K. (2025). Human Wound and Its Burden: Updated 2025 Compendium of Estimates. Advances in wound care, 14(9), 429–438.
- Iversen, A. K. S., Lichtenberg, M., Fritz, B. G., Díaz-Pinés Cort, I., Al-Zoubaidi, D. F., Gottlieb, H., Kirketerp-Møller, K., Bjarnsholt, T., & Jakobsen, T. H. (2024). The chronic wound characterisation study and biobank: a study protocol for a prospective observational cohort investigation of bacterial community composition, inflammatory responses and wound-healing trajectories in non-healing wounds. BMJ open, 14(10), e084081.
- Martinengo, L., Olsson, M., Bajpai, R., Soljak, M., Upton, Z., Schmidtchen, A., Car, J., & Järbrink, K. (2019). Prevalence of chronic wounds in the general population: systematic review and meta-analysis of observational studies. Annals of epidemiology, 29, 8–15.
- Carter, M. J., DaVanzo, J., Haught, R., Nusgart, M., Cartwright, D., & Fife, C. E. (2023). Chronic wound prevalence and the associated cost of treatment in Medicare beneficiaries: changes between 2014 and 2019. Journal of medical economics, 26(1), 894–901.
- Queen, D., & Harding, K. (2024). Estimating the cost of wounds both nationally and regionally within the top 10 highest spenders. International wound journal, 21(2), e14709.
- Silini, A. R., Cargnoni, A., Magatti, M., Pianta, S., & Parolini, O. (2015). The Long Path of Human Placenta, and Its Derivatives, in Regenerative Medicine. Frontiers in bioengineering and biotechnology, 3, 162.
- Järbrink, K., Ni, G., Sönnergren, H., Schmidtchen, A., Pang, C., Bajpai, R., & Car, J. (2016). Prevalence and incidence of chronic wounds and related complications: a protocol for a systematic review. Systematic reviews, 5(1), 152.
- Protzman, N. M., Mao, Y., Long, D., Sivalenka, R., Gosiewska, A., Hariri, R. J., & Brigido, S. A. (2023). Placental-Derived Biomaterials and Their Application to Wound Healing: A Review. Bioengineering, 10(7), 829.
- Singh, P., Easley, A., Menchaca, K. T., Fanniel, V., Gomez, R., Marquez, J., & Hill, S. (2025). Comparative Study of Placental Allografts with Distinct Layer Composition. International Journal of Molecular Sciences, 26(7), 3406.
- Thowfeequ, S., & Srinivas, S. (2022). Embryonic and extraembryonic tissues during mammalian development: shifting boundaries in time and space. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 377(1865), 20210255.
- Menon, R., & Moore, J. J. (2020). Fetal Membranes, Not a Mere Appendage of the Placenta, but a Critical Part of the Fetal-Maternal Interface Controlling Parturition. Obstetrics and gynecology clinics of North America, 47(1), 147–162.
- Menon, R., Richardson, L. S., & Lappas, M. (2019). Fetal membrane architecture, aging and inflammation in pregnancy and parturition. Placenta, 79, 40–45.
- Rouzaire, M., Blanchon, L., Sapin, V., & Gallot, D. (2024). Application of Fetal Membranes and Natural Materials for Wound and Tissue Repair. International Journal of Molecular Sciences, 25(22), 11893.
- A. Roy, M. Mantay, C. Brannan, and S. Griffiths, "Placental Tissues as Biomaterials in Regenerative Medicine”, BioMed Research International, 2022, 6751456, 26 pages, 2022.
- L. Richardson and R. Menon, "Fetal membrane at the feto-maternal interface: An underappreciated and understudied intrauterine tissue," Placenta and Reproductive Medicine, vol. 1, Nov. 2022,
- R. Menon, M. Lappas, and T. Zakar, "Editorial: The Role of the Fetal Membranes in Pregnancy and Birth," Feb. 18, 2021, Frontiers Media S.A.
- Z. Hu et al., "Biological importance of human amniotic membrane in tissue engineering and regenerative medicine," Oct. 01, 2023, Elsevier B.V.
- M. Sabbatini, P. Boffano, M. Ferrillo, M. Migliario, and F. Renò, "The Human Amniotic Membrane: A Rediscovered Tool to Improve Wound Healing in Oral Surgery," Sep. 01, 2025, Multidisciplinary Digital Publishing Institute (MDPI).
- A. C. Mamede, M. J. Carvalho, A. M. Abrantes, M. Laranjo, C. J. Maia, and M. F. Botelho, "Amniotic membrane: From structure and functions to clinical applications," Aug. 01, 2012, Springer Verlag.
- H. S. Dua, J. A. P. Gomes, A. J. King, and V. S. Maharajan, "The amniotic membrane in ophthalmology," Surv. Ophthalmol., vol. 49, no. 1, pp. 51–77, 2004,
- B. J. Van Herendael, C. Oberti, and I. Brosens, "Microanatomy of the human amniotic membrane. A light microscopic, transmission, and scanning electron microscopic study," Am. J. Obstet. Gynecol., vol. 131, no. 8, pp. 872–880, 1978,
- C. T. Koh, K. Tonsomboon, and M. L. Oyen, "Fracture toughness of human amniotic membranes," Interface Focus, vol. 9, no. 5, Oct. 2019,
- R. Mohan, A. Bajaj, and M. Gundappa, "Human amnion membrane: Potential applications in oral and periodontal field," Jan. 01, 2017, Wolters Kluwer (UK) Ltd.
- A. Toda, M. Okabe, T. Yoshida, and T. Nikaido, "The potential of amniotic membrane/amnion-derived cells for regeneration of various tissues," 2007, Japanese Pharmacological Society.
- C. Malhotra and A. K. Jain, "Human amniotic membrane transplantation: Different modalities of its use in ophthalmology," World J. Transplant., vol. 4, no. 2, p. 111, 2014,
- B. Farhadihosseinabadi et al., "Amniotic membrane and its epithelial and mesenchymal stem cells as an appropriate source for skin tissue engineering and regenerative medicine," Nov. 05, 2018, Taylor and Francis Ltd.
- J. W. Kang et al., "Immunomodulatory effects of human amniotic membrane-derived mesenchymal stem cells," J. Vet. Sci., vol. 13, no. 1, pp. 23–31, 2012,
- L. Zhang et al., "An Ultra-thin Amniotic Membrane as Carrier in Corneal Epithelium Tissue-Engineering," Sci. Rep., vol. 6, Feb. 2016,
- HAMILTON, W. J., & BOYD, J. D. (1960). Development of the human placenta in the first three months of gestation. Journal of anatomy, 94(Pt 3), 297–328
- S. Ilancheran, Y. Moodley, and U. Manuelpillai, "Human Fetal Membranes: A Source of Stem Cells for Tissue Regeneration and Repair?," Placenta, vol. 30, no. 1, pp. 2–10, Jan. 2009,
- S. W. Verbruggen, M. L. Oyen, A. T. M. Phillips, and N. C. Nowlan, "Function and failure of the fetal membrane: Modelling the mechanics of the chorion and Amnion," PLoS One, vol. 12, no. 3, Mar. 2017,
- R. Menon, L. S. Richardson, and M. Lappas, "Fetal membrane architecture, aging and inflammation in pregnancy and parturition," Apr. 01, 2019, W.B. Saunders Ltd.
- M. L. Oyen, S. E. Calvin, and D. V. Landers, "Premature rupture of the fetal membranes: Is the amnion the major determinant?," Am. J. Obstet. Gynecol., vol. 195, no. 2, pp. 510–515, Aug. 2006,
- H. Oxlund ', R. Helmig, J. T. Halaburt, and N. Uldbjerg, "Biomechanical analysis of human chorioamniotic membranes," 1990
- G. Bourne, M. R. C. O. G. Obstetric, G. Surgeon, S. Bartholomew's Hospital, and E. C. I. London, "The Foetal Membranes A Review of the Anatomy of Normal Amnion and Chorion and Some Aspects of Their Function," 1962
- Lei, Jennifer; Priddy, Lauren B.; Lim, Jeremy J.; Koob, Thomas J. Dehydrated Human Amnion/Chorion Membrane (dHACM) Allografts as a Therapy for Orthopedic Tissue Repair. Techniques in Orthopaedics 32(3):p 149-157, September 2017. DOI: 10.1097/BTO.0000000000000229
- R. Menon, E. Radnaa, F. Behnia, and R. Urrabaz-Garza, "Isolation and characterization human chorion membrane trophoblast and mesenchymal cells," Placenta, vol. 101, pp. 139–146, Nov. 2020,
- Van Meir, C. A., Sangha, R. K., Walton, J. C., Matthews, S. G., Keirse, M. J., & Challis, J. R. (1996). Immunoreactive 15-hydroxyprostaglandin dehydrogenase (PGDH) is reduced in fetal membranes from patients at preterm delivery in the presence of infection. Placenta, 17(5-6), 291–297.
- Y. Ogawa et al., "Heavy Chain-Hyaluronan/Pentraxin 3 from Amniotic Membrane Suppresses Inflammation and Scarring in Murine Lacrimal Gland and Conjunctiva of Chronic Graft-versus-Host Disease," Sci. Rep., vol. 7, Feb. 2017,
- S. C. G. Tseng, "HC-HA/PTX3 purified from amniotic membrane as novel regenerative matrix: Insight into relationship between inflammation and regeneration," Invest. Ophthalmol. Vis. Sci., vol. 57, no. 5, pp. ORSFh1–ORSFh8, Apr. 2016.
- S. C. G. Tseng et al., "How Does Amniotic Membrane Work? Laboratory Science," 2004
- H. Niknejad, H. Peirovi, M. Jorjani, A. Ahmadiani, J. Ghanavi, and A. M. Seifalian, "Properties of the amniotic membrane for potential use in tissue engineering," 2008, AO Research Institute Davos.
- E. Guzeldemir-Akcakanat, D. Kan-Karabiyik, and D. Yigit, "The Human Amniotic Membrane and its Applications in Regenerative Dentistry: An Overview," Dec. 01, 2025, Springer Science and Business Media B.V.
- M. Gholipourmalekabadi, B. Farhadihosseinabadi, M. Faraji, and M. R. Nourani, "How preparation and preservation procedures affect the properties of amniotic membrane? How safe are the procedures?," Sep. 01, 2020, Elsevier Ltd.
- G. Dawiec, W. Niemczyk, R. Wiench, S. Niemczyk, and D. Skaba, "Introduction to Amniotic Membranes in Maxillofacial Surgery—A Scoping Review," Apr. 01, 2024, Multidisciplinary Digital Publishing Institute (MDPI).
- T. Ito, J. D. Williams, D. J. Fraser, and A. O. Phillips, "Hyaluronan regulates transforming growth factor-β1 receptor compartmentalization," Journal of Biological Chemistry, vol. 279, no. 24, pp. 25326–25332, Jun. 2004,
- H. He et al., "Biochemical characterization and function of complexes formed by hyaluronan and the heavy chains of inter-α-inhibitor (HC·HA) purified from extracts of human amniotic membrane," Journal of Biological Chemistry, vol. 284, no. 30, pp. 20136–20146, Jul. 2009.
- M. S. Lord, F. Tang, J. Rnjak-Kovacina, J. G. W. Smith, J. Melrose, and J. M. Whitelock, "The multifaceted roles of perlecan in fibrosis," Matrix Biology, vol. 68–69, pp. 150–166, Aug. 2018,
- S. A. Antar, N. A. Ashour, M. E. Marawan, and A. A. Al-Karmalawy, "Fibrosis: Types, Effects, Markers, Mechanisms for Disease Progression, and Its Relation with Oxidative Stress, Immunity, and Inflammation," Feb. 01, 2023, Multidisciplinary Digital Publishing Institute (MDPI).
- C. H. Wassmer and E. Berishvili, "Immunomodulatory Properties of Amniotic Membrane Derivatives and Their Potential in Regenerative Medicine," Aug. 01, 2020, Springer.
- L. B. Sant'Anna et al., "Antifibrotic effects of human amniotic membrane transplantation in established biliary fibrosis induced in rats," Cell Transplant., vol. 25, no. 12, pp. 2245–2257, 2016,
- S. E. Moreno, M. Massee, and T. J. Koob, "Dehydrated Human Amniotic Membrane Inhibits Myofibroblast Contraction through the Regulation of the TGFβ‒SMAD Pathway In Vitro," JID Innov., vol. 1, no. 2, Jun. 2021
- G. L. Bourne, "The microscopic anatomy of the human amnion and chorion," Am. J. Obstet. Gynecol., vol. 79, no. 6, pp. 1070–1073, 1960,
- M. K. Arki, K. Moeinabadi-Bidgoli, N. Hossein-Khannazer, R. Gramignoli, M. Najimi, and M. Vosough, "Amniotic Membrane and Its Derivatives: Novel Therapeutic Modalities in Liver Disorders," Aug. 01, 2023, Multidisciplinary Digital Publishing Institute (MDPI).
- S. Wolbank, F. Hildner, H. Redl, M. Van Griensven, C. Gabriel, and S. Hennerbichler, "Impact of human amniotic membrane preparation on release of angiogenic factors," J. Tissue Eng. Regen. Med., vol. 3, no. 8, pp. 651–654, 2009,
- A. Hopkinson, R. S. McIntosh, P. J. Tighe, D. K. James, and H. S. Dua, "Amniotic membrane for ocular surface reconstruction: Donor variations and the effect of handling on TGF-β content," Invest. Ophthalmol. Vis. Sci., vol. 47, no. 10, pp. 4316–4322, Oct. 2006,
- R. W. D. Gilbert, M. K. Vickaryous, and A. M. Viloria-Petit, "Signalling by transforming growth factor beta isoforms in wound healing and tissue regeneration," Jun. 01, 2016, MDPI Multidisciplinary Digital Publishing Institute
- S.-B. Lee, D.-Q. Li, D. Tan, D. Meller, and S. C. G. Tseng, "Lee2000SuppressionofTGF-bsignalinginbothnormalconjunctivalfibroblastsandpterygialbodyfibroblasts," Curr. Eye Res., vol. 20, pp. 325–334, Mar. 2000
- H. Elkhenany, A. El-Derby, M. Abd Elkodous, R. A. Salah, A. Lotfy, and N. El-Badri, "Applications of the amniotic membrane in tissue engineering and regeneration: the hundred-year challenge," Dec. 01, 2022, BioMed Central Ltd.
- R. P. Galask and I. S. Snyder, "Antimicrobial factors in amniotic fluid," Am. J. Obstet. Gynecol., vol. 106, no. 1, pp. 59–65, Jan. 1970.
- Y. P. TALMIb, L. Sigler, E. Inge, Y. Finkelstein, and Y. Zohar, "Antibacterial Membranes Properties of Human Amniotic," 1991
- Y. Mao et al., "Antimicrobial Peptides Secreted from Human Cryopreserved Viable Amniotic Membrane Contribute to its Antibacterial Activity," Sci. Rep., vol. 7, no. 1, Dec. 2017,
- Kanyshkova, T. G., Buneva, V. N., & Nevinsky, G. A. (2001). Lactoferrin and its biological functions. Biochemistry (Moscow), 66(1), 1–7.
- Eskandarlou, M., Azimi, M., Rabiee, S., & Seif Rabiee, M. A. (2016). The Healing Effect of Amniotic Membrane in Burn Patients. World journal of plastic surgery, 5(1), 39–44.
- M. G. Tonnesen, X. Feng, and R. A. F. Clark, "Angiogenesis in wound healing," Journal of Investigative Dermatology Symposium Proceedings, vol. 5, no. 1, pp. 40–46, 2000.
- Nishida, N., Yano, H., Nishida, T., Kamura, T., and Kojiro, M. (2006). Angiogenesis in cancer. Vascular health and risk management, 2(3), 213–219.
- Z. Grzywocz et al., "Growth factors and their receptors derived from human amniotic cells in vitro," Folia Histochem. Cytobiol., vol. 52, no. 3, pp. 163–170, 2014,
- Y. M. D. , M. S. ; M. D. H.-K. M. D. ; H. D. G. M. D. ; K. W.-S. M. D. ,; Z. F.. Hao, "Identification of Antiangiogenic and Antiinflammatory Proteins in Human Amniotic Membrane," Cornea, pp. 348–352, 2000
- Z. L. Liu, H. H. Chen, L. L. Zheng, L. P. Sun, and L. Shi, "Angiogenic signaling pathways and anti-angiogenic therapy for cancer," Dec. 01, 2023, Springer Nature.
- H. Niknejad, G. Paeini-Vayghan, F. A. Tehrani, M. Khayat-Khoei, and H. Peirovi, "Side dependent effects of the human amnion on angiogenesis," Placenta, vol. 34, no. 4, pp. 340–345, Apr. 2013,
- L. S. Lentz et al., "Human chorionic gonadotropin promotes murine Treg cells and restricts pregnancy-harmful proinflammatory Th17 responses," Front. Immunol., vol. 13, Sep. 2022,
- M. Silasi et al., "Human Chorionic Gonadotropin modulates CXCL10 Expression through Histone Methylation in human decidua," Sci. Rep., vol. 10, no. 1, Dec. 2020.
- O. A. G. Tantengco et al., "Histocompatibility antigen, class i, g (Hla-g)'s role during pregnancy and parturition: A systematic review of the literature," Oct. 01, 2021, MDPI.
- C. Megli, S. Morosky, D. Rajasundaram, and C. B. Coyne, "Inflammasome signaling in human placental trophoblasts regulates immune defense against Listeria monocytogenes infection," Journal of Experimental Medicine, vol. 218, no. 1, 2021,
- S. S. Burgener and K. Schrode, "Placental inflammasome signaling: Protection for mother and baby," Journal of Experimental Medicine, vol. 218, no. 1, Nov. 2020,
- Uchide, N., Ohyama, K., Bessho, T., & Toyoda, H. (2005). Induction of pro-inflammatory cytokine gene expression and apoptosis in human chorion cells of fetal membranes by influenza virus infection: Possible implications for maintenance and interruption of pregnancy during infection. Medical Science Monitor, 11(1), RA7–RA16.
- T. Habelrih et al., "Inflammatory mechanisms of preterm labor and emerging anti-inflammatory interventions," Aug. 01, 2024, Elsevier Ltd.
- S. Shariatzadeh et al., "Developing a pro-angiogenic placenta derived amniochorionic scaffold with two exposed basement membranes as substrates for cultivating endothelial cells," Sci. Rep., vol. 11, no. 1, Dec. 2021.
- N. Pavlov, J. L. Frendo, J. Guibourdenche, S. A. Degrelle, D. Evain-Brion, and J. Badet, "Angiogenin expression during early human placental development; Association with blood vessel formation," Biomed Res. Int., vol. 2014, 2014,
- C. C. Bray, C. M. Walker, and D. D. Spence, "Orthobiologics in Pediatric Sports Medicine," Orthopedic Clinics of North America, vol. 48, no. 3, pp. 333–342, Jul. 2017.
- M. A. Moore, B. Samsell, and J. McLean, "Allograft tissue safety and technology," in Biologics in Orthopaedic Surgery, Elsevier, 2018, pp. 49–62.
- A. Klama-Baryła et al., "Preparation of placental tissue transplants and their application in skin wound healing and chosen skin bullous diseases - Stevens-Johnson syndrome and toxic epidermal necrolysis treatment," Int. Wound J., vol. 17, no. 2, pp. 491–507, Apr. 2020.
- T. H. Jang et al., "Cryopreservation and its clinical applications," Integr. Med. Res., vol. 6, no. 1, pp. 12–18, Mar. 2017.
- Kruse, F. E., Rohrschneider, K., & Völcker, H. E. (2000). Cryopreserved human amniotic membrane for ocular surface reconstruction. Graefe’s Archive for Clinical and Experimental Ophthalmology, 238(1), 68–75.
- M. T. Rodríguez-Ares et al., "Effects of lyophilization on human amniotic membrane," Acta Ophthalmol., vol. 87, no. 4, pp. 396–403, Jun. 2009.
- M. E. McCarthy et al., "Decellularization of Human Digits: A Step Towards Off-the-Shelf Composite Allograft Transplantation," Bioengineering, vol. 12, no. 4, Apr. 2025.
- M. Gadre, M. Kasturi, P. Agarwal, and K. S. Vasanthan, "Decellularization and Their Significance for Tissue Regeneration in the Era of 3D Bioprinting," 2023, American Chemical Society.
- G. Matus, J. P. Aravena, D. Mariño, and S. E. Niklander, "Decellularized allografts as an alternative for reconstruction of large inferior alveolar nerve defects: a systematic review," 2023, Medicina Oral S.L.
- J. M. Aamodt and D. W. Grainger, "Extracellular matrix-based biomaterial scaffolds and the host response," Apr. 01, 2016, Elsevier Ltd.
- M. Kasravi et al., "Immunogenicity of decellularized extracellular matrix scaffolds: a bottleneck in tissue engineering and regenerative medicine," Dec. 01, 2023, BioMed Central Ltd.
- Z. Zhang, B. Lu, L. Zou, X. Huang, F. Yang, and H. Lv, "Decellularized human amniotic membrane scaffolds: influence on the biological behavior of dental pulp stem cells," BMC Oral Health, vol. 24, no. 1, Dec. 2024.
- L. S. Lim, R. W. Y. Poh, A. K. Riau, R. W. Beuerman, D. Tan, and J. S. Mehta, "Biological and Ultrastructural Properties of Acelagraft, a Freeze-Dried-Irradiated Human Amniotic Membrane," 2010
- F. von Versen-Hoeynck, A. P. Steinfeld, J. Becker, M. Hermel, W. Rath, and U. Hesselbarth, "Sterilization and preservation influence the biophysical properties of human amnion grafts," Biologicals, vol. 36, no. 4, pp. 248–255, Jul. 2008.
- R. Singh, D. Singh, and A. Singh, "Radiation sterilization of tissue allografts: A review," World J. Radiol., vol. 8, no. 4, p. 355, 2016.
- F. Von Versen-Höynck, C. Syring, S. Bachmann, and D. E. Möller, "The influence of different preservation and sterilisation steps on the histological properties of amnion allografts-light and scanning electron microscopic studies," 2004
- H. Suroto, D. M. Aryawan, and C. A. Prakoeswa, "The Influence of the Preservation Method and Gamma Irradiation Sterilization on TGF- β and bFGF Levels in Freeze-Dried Amnion Membrane (FD-AM) and Amnion Sponge," Int. J. Biomater., vol. 2021.
- J. O'Connell et al., "Development of a sterilization process for amniotic membrane allograft tissue using supercritical carbon dioxide and NovaKill," Cell Tissue Bank., vol. 26, no. 1, Mar. 2025,
- L. Becktell et al., "Proteomic Analysis and Cell Viability of Nine Amnion, Chorion, Umbilical Cord, and Amniotic Fluid–Derived Products," Dec. 01, 2021, SAGE Publications Inc.
- S. Barrientos, O. Stojadinovic, M. S. Golinko, H. Brem, and M. Tomic-Canic, "Growth factors and cytokines in wound healing," Sep. 2008.
- T. J. Koob, J. J. Lim, N. Zabek, and M. Massee, "Cytokines in single layer amnion allografts compared to multilayer amnion/chorion allografts for wound healing," J. Biomed. Mater. Res. B Appl. Biomater., vol. 103, no. 5, pp. 1133–1140, Jul. 2015.
- McQuilling, J. P., Vines, J. B., Kimmerling, K. A., & Mowry, K. C. (2017). Proteomic comparison of amnion and chorion and evaluation of the effects of processing on placental membranes. Wounds, 29(6), E36–E40.
- T. G. Kim, K. Do Ki, M. K. Lee, J. W. So, S. K. Chung, and J. Kang, "Comparison of cytokine expression and ultrastructural alterations in fresh-frozen and dried electron beam-irradiated human amniotic membrane and chorion," Cell Tissue Bank., vol. 20, no. 2, pp. 163–172, Jun. 2019.
- T. J. Sabol, G. S. Tran, J. Matuszewski, and W. W. Weston, "Standardized reporting of amnion and amnion/chorion allograft data for wound care," Health Sci. Rep., vol. 5, no. 5, Sep. 2022.
- M. Riaz, M. Z. Iqbal, A. S. Klar, and T. Biedermann, "Immunomodulatory Mechanisms of Chronic Wound Healing: Translational and Clinical Relevance," Nov. 01, 2025, John Wiley and Sons Inc.
- M. P. Caley, V. L. C. Martins, and E. A. O'Toole, "Metalloproteinases and Wound Healing," Adv. Wound Care (New. Rochelle)., vol. 4, no. 4, pp. 225–234, Apr. 2015,
- O. S. Folorunso et al., "Tissue Inhibitor of Metalloproteinase 2 Promotes Wound Healing by Suppressing Matrix Metalloproteinases and Inflammatory Cytokines in Corneal Epithelial Cells," Am. J. Pathol., vol. 195, no. 4, pp. 754–769, Apr. 2025,
- S. Moreno, M. Massee, S. Campbell, H. Bara, T. J. Koob, and J. R. Harper, "PURION® processed human amnion chorion membrane allografts retain material and biological properties supportive of soft tissue repair," J. Biomater. Appl., vol. 39, no. 1, pp. 24–39, Jul. 2024,
- P. Pfister et al., "Human amniotic membranes as an allogenic biological dressing for the treatment of burn wounds: Protocol for a randomized-controlled study," Contemp. Clin. Trials Commun., vol. 36, Dec. 2023,
- N. G. Fairbairn, M. A. Randolph, and R. W. Redmond, "The clinical applications of human amnion in plastic surgery," 2014, Churchill Livingstone.
- A. Garten, L. M. Nherera, and R. Lindsay, "Comparison of Single Use and Traditional Negative Pressure Wound Therapy Devices in Lower Extremity Ulcers: A US Real-World Evidence Analysis of NetHealth Data," Int. Wound J., vol. 22, no. 9, Sep. 2025.
- K. McDermott, M. Fang, A. J. M. Boulton, E. Selvin, and C. W. Hicks, "Etiology, Epidemiology, and Disparities in the Burden of Diabetic Foot Ulcers," Jan. 01, 2023, American Diabetes Association Inc.
- S. F. Bernatchez, J. Eysaman-Walker, and D. Weir, "Venous Leg Ulcers: A Review of Published Assessment and Treatment Algorithms," Adv. Wound Care (New. Rochelle)., vol. 11, no. 1, pp. 28–41, Jan. 2022,
- J. M. Raja, M. A. Maturana, S. Kayali, A. Khouzam, and N. Efeovbokhan, "Diabetic foot ulcer: A comprehensive review of pathophysiology and management modalities," World J. Clin. Cases, vol. 11, no. 8, pp. 1684–1693, Mar. 2023.
- J. Dawi et al., "Diabetic Foot Ulcers: Pathophysiology, Immune Dysregulation, and Emerging Therapeutic Strategies," May 01, 2025, Multidisciplinary Digital Publishing Institute (MDPI).
- J. D. Raffetto, D. Ligi, R. Maniscalco, R. A. Khalil, and F. Mannello, "Why venous leg ulcers have difficulty healing: Overview on pathophysiology, clinical consequences, and treatment," Jan. 01, 2021, MDPI.
- J. Kim, O. Nomkhondorj, C. Y. An, Y. C. Choi, and J. Cho, "Management of diabetic foot ulcers: a narrative review," Oct. 01, 2023, Yeungnam University School of Medicine and College of Medicine.
- J. Caporusso et al., "A Multi-Centre, Randomised, Controlled Clinical Trial Assessing Cryopreserved Ultra-Thick Human Amniotic Membrane in the Treatment of Complex Diabetic Foot Ulcers," Wound Repair and Regeneration, vol. 33, no. 6, Nov. 2025.
- C. Becker, M. Regulski, S. Martin, and T. Barrett, "Application of Dehydrated Amniotic Membrane Allografts in Advanced Diabetic Foot Ulceration: Case Report and Review of Literature," Reports, vol. 5, no. 3, p. 28, Jul. 2022.
- J. R. Hanft, T. Tucker, and T. Gillespie, "Comparative evaluation of skin substitutes for diabetic foot ulcers and venous leg ulcers using the Medicare Administrative Claims Database," International Journal of Tissue Repair, vol. 1, no. 1, 2025.
- A. M. Haugh et al., "Amnion membrane in diabetic foot wounds: A meta-analysis," Plast. Reconstr. Surg. Glob. Open, vol. 5, no. 4, 2017.
- C. Zheng, W. Tang, and X. Ran, "Efficacy and Safety of Human Amniotic Membrane for Chronic Wounds: A Systematic Review and Meta-Analysis of Clinical Trials," Adv. Wound Care (New. Rochelle)., Jan. 2026.
- F. Game et al., "The effectiveness of a new dried human amnion derived membrane in addition to standard care in treating diabetic foot ulcers: A patient and assessor blind, randomised controlled pilot study," Int. Wound J., vol. 18, no. 5, pp. 692–700, Oct. 2021.
- S. M. Barr, "Dehydrated Amniotic Membrane Allograft for Treatment of Chronic Leg Ulcers in Patients With Multiple Comorbidities: A Case Series," Journal of the American College of Clinical Wound Specialists, vol. 6, no. 3, pp. 38–45, Dec. 2014.
- R. D. Galiano, D. P. Orgill, D. G. Armstrong, P. M. Glat, M. J. Carter, and C. M. Zelen, "A Prospective Multicenter Study of a Weekly Application Regimen of Viable Human Amnion Membrane Allograft in the Management of Nonhealing Diabetic Foot Ulcers," Plast. Reconstr. Surg. Glob. Open, vol. 11, no. 10, p. E5291, Oct. 2023.
- Y. Terabe and N. Kaneko, "Effectivity of the Dehydrated Human Amnion/Chorion Membrane Allograft for Diabetic Foot Ulcers in Japan: A Case Series," Int. Wound J., vol. 22, no. 11, Nov. 2025.
- C. M. Zelen, T. E. Serena, and D. E. Fetterolf, "Dehydrated human amnion/chorion membrane allografts in patients with chronic diabetic foot ulcers: A long-term follow-up study," Wound Medicine, vol. 4, pp. 1–4, 2014,
- T. E. Serena et al., "A multicenter, randomized, controlled clinical trial evaluating the use of dehydrated human amnion/chorion membrane allografts and multilayer compression therapy vs. multilayer compression therapy alone in the treatment of venous leg ulcers," Wound Repair and Regeneration, vol. 22, no. 6, pp. 688–693, Nov. 2014,
- T. E. Serena et al., "A Multicenter, Randomized, Controlled, Clinical Trial Evaluating Dehydrated Human Amniotic Membrane in the Treatment of Venous Leg Ulcers," Plast. Reconstr. Surg., vol. 150, no. 5, pp. 1128–1136, Nov. 2022,
- A. Oropallo, A. Goodwin, M. K. Morrissey, C. Del Pin, and A. Rao, "Human Amnion Chorion Membrane Allografts in the Treatment of Chronic Diabetic Foot Ulcers: A Literature Review," Apr. 01, 2021, Lippincott Williams and Wilkins.
- W. Tettelbach, S. Cazzell, A. M. Reyzelman, F. Sigal, J. M. Caporusso, and P. S. Agnew, "A confirmatory study on the efficacy of dehydrated human amnion/chorion membrane dHACM allograft in the management of diabetic foot ulcers: A prospective, multicentre, randomised, controlled study of 110 patients from 14 wound clinics," Int. Wound J., vol. 16, no. 1, pp. 19–29, Feb. 2019,
- C. Bianchi, S. Cazzell, D. Vayser, A. M. Reyzelman, H. Dosluoglu, and G. Tovmassian, "A multicentre randomised controlled trial evaluating the efficacy of dehydrated human amnion/chorion membrane (EpiFix®) allograft for the treatment of venous leg ulcers," Int. Wound J., vol. 15, no. 1, pp. 114–122, Feb. 2018.
- Cazzell, S. M., Caporusso, J., Vayser, D., Davis, R. D., Alvarez, O. M., & Sabolinski, M. L. (2024). Dehydrated amnion chorion membrane versus standard of care for diabetic foot ulcers: A randomised controlled trial. Journal of Wound Care, 33(Sup7), S4–S14.
- J. Paris, C. Macri, A. I. Almater, J. Slattery, and D. Selva, "Amniotic membrane transplantation techniques in acute ocular Stevens-Johnson syndrome and toxic epidermal necrolysis: A systematic review," Oct. 01, 2025, Elsevier Inc.
- I. F. Baig, N. T. Le, and Z. Al-Mohtaseb, "Amniotic membrane transplantation: an updated clinical review for the ophthalmologist," 2023, AME Publishing Company.
- N. Hofmann, H. O. Rennekampff, A. K. Salz, and M. Börgel, "Preparation of human amniotic membrane for transplantation in different application areas," 2023, Frontiers Media SA.
- A. L. Ingraldi, R. G. Audet, and A. J. Tabor, "The Preparation and Clinical Efficacy of Amnion-Derived Membranes: A Review," Oct. 01, 2023, Multidisciplinary Digital Publishing Institute (MDPI).
- S. H. Salehi, K. As'adi, S. J. Mousavi, and S. Shoar, "Evaluation of Amniotic Membrane Effectiveness in Skin Graft Donor Site Dressing in Burn Patients," Indian Journal of Surgery, vol. 77, pp. 427–431, Dec. 2015.
- Kadkhoda, Z., Tavakoli, A., Chokami Rafiei, S., Zolfaghari, F., & Akbari, S. (2020). Effect of amniotic membrane dressing on pain and healing of palatal donor site: A randomized controlled trial. International Journal of Organ Transplantation Medicine, 11(2), 55–62.
- S. Thatte and J. Jain, "Fornix reconstruction with amniotic membrane transplantation: A cosmetic remedy for blind patients," J. Ophthalmic Vis. Res., vol. 11, no. 2, pp. 193–197, Apr. 2016,
- G. Leila et al., "A randomized trial study on the effect of amniotic membrane graft on wound healing process after anal fistulotomy," Journal of Coloproctology, vol. 37, no. 3, pp. 187–192, Jul. 2017,
- Garcia, N., Jiminez, V., Graham, L., & Huang, C. (2023). Unique usages of dehydrated human amnion chorion membrane allografts in dermatology. Journal of Drugs in Dermatology, 22(12), 1228–1231.
- S. Odet et al., "Surgical Application of Human Amniotic Membrane and Amnion-Chorion Membrane in the Oral Cavity and Efficacy Evaluation: Corollary With Ophthalmological and Wound Healing Experiences," Jun. 10, 2021, Frontiers Media S.A.
- S. Kogan, A. Sood, and M. S. Granick, "Amniotic Membrane Adjuncts and Clinical Applications in Wound Healing: A Review of the Literature.," Wounds, vol. 30, no. 6, pp. 168–173, Jun. 2018
- Choat, D., Das, B., & Bardin, M. (2025). Clinical outcomes utilizing a dehydrated human amnion/chorion membrane (DHACM) allograft as a protective barrier against colorectal anastomotic leaks in patients with inflammatory bowel disease (IBD). Inflammatory Bowel Diseases, 31(Suppl 1), S6.
- P. B. Tonape, J. V. S. Kishore, R. M. Kopparthi, T. Tonape, D. S. Bhamare, and S. Desireddy, "Clinico-radiological outcome of Arthroscopic Anterior Cruciate Ligament Reconstruction with Augmentation of Dehydrated Human Amnion Chorion Allograft Membrane using Peroneus Longus Autograft," Malays. Orthop. J., vol. 18, no. 1, pp. 33–41, Mar. 2024.
- Gellhorn, A. C., & Han, A. (2017). The use of dehydrated human amnion/chorion membrane allograft injection for the treatment of tendinopathy or arthritis: A case series involving 40 patients. PM&R, 9(12), 1236–1243.
- B. R. Subach and A. G. Copay, "The Use of a Dehydrated Amnion/Chorion Membrane Allograft in Patients Who Subsequently Undergo Reexplanation after Posterior Lumbar Instrumentation," Adv. Orthop., vol. 2015, 2015.
 Alcrut
Alcrut