Review Article
1 Staysis Medical, Scottsdale, Arizona, USA.
2 Aaron J. Tabor, Medtech Incubator, Scottsdale, Arizona, USA.
*Corresponding Author: Aaron J. Tabor, Medtech Incubator, Scottsdale, Arizona, USA.
Citation: Bryce Jewett and Aaron Tabor, Demineralized Bone Matrix and Cellular Bone Allografts: Mechanistic, Clinical, and Translational Comparisons Across Commercial Platforms, J Clinical and Medical Research and Studies, V (5)I(2), DOI: 10.59468/2836-8525/152
Copyright: 2026 Aaron J. Tabor. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: March 30, 2026 | Accepted: April 03, 2026 | Published: April 04, 2026
Abstract
Demineralized bone matrix (DBM) and cellular bone allografts (CBA) represent two widely utilized osteobiologic strategies in spinal fusion and orthopedic reconstruction. While DBM primarily provides osteoinductive growth factors within a demineralized collagen scaffold, CBAs incorporate viable mesenchymal stem or progenitor cells in addition to structural matrix components. Despite increasing clinical adoption of both graft categories, comparative evaluation across mechanistic, clinical, regulatory, and economic domains remains limited. Accordingly, the current evidence does not support the superiority of any specific graft type across different clinical applications. This review proposes a structured translational framework for comparative graft evaluation integrating biologic mechanism, clinical performance, regulatory context, and economic considerations.
Keywords: Demineralized bone matrix (DBM); cellular bone allografts (CBA); bone healing, osteogenesis; osteoinduction, and osteoconduction
1. Introduction
Bone grafting remains one of the most commonly performed procedures in orthopedic and spinal surgery, with applications spanning spinal fusion, fracture reconstruction, and the management of osseous defects arising from developmental onset, trauma, revision surgery, or degenerative disease. The volume and complexity of these procedures have increased substantially over recent decades, driven in part by an aging population, broadened surgical indications, and advances in fixation technology [135, 164]. As the demand for bone grafting has grown, so too has the need for reliable, well-characterized graft materials capable of supporting predictable bone regeneration across a range of clinical contexts.
Autologous bone graft, most commonly harvested from the iliaccrest, has long been regarded as the reference standard for bone grafting because it provides all three components of the osteogenic triad: osteogenic cells, osteoinductive signaling molecules, and an osteoconductive scaffold [104, 124].However, autograft use is subject to several well-documented limitations that constrain its clinical utility. Donor-site morbidity, including chronic pain, hematoma, infection, and nerve injury, has been reported in a substantial proportion of patients undergoing iliac crest bone graft harvest [114, 115]. Available graft volume is finite and may be insufficient for large or multilevel surgical constructs, particularly in revision settings. Additionally, the biological quality of autograft tissue may vary with patient age, metabolic status, and comorbid conditions, introducing further uncertainty regarding its osteogenic reliability. These limitations have motivated the development and clinical adoption of a diverse array of bone graft substitutes and alternatives intended to reduce dependence on autologous harvest while maintaining acceptable healing outcomes [87, 165].
These limitations have driven a progression in graft substitute development that broadly follows the expanding understanding of bone biology. Early alternatives focused on providing osteoconductive scaffolding and preserving matrix-bound growth factors through demineralization of cadaveric bone, yielding the category now recognized as demineralized bone matrix. As the role of viable progenitor cells in bone repair became better characterized, a subsequent generation of graft technologies sought to incorporate living cellular populations within allogeneic matrices, giving rise to cellular bone allografts. More recently, recombinant growth factor platforms, synthetic scaffolds with bioactive modifications, and emerging cell-based therapies have further expanded the landscape of commercially available options. This trajectory from passive scaffold to biologically augmented construct reflects both genuine scientific advancement and the commercial imperative to differentiate products within an increasingly competitive market, and it provides the context within which the comparative analysis that follows should be understood.
Among the most widely utilized categories of commercially available bone graft substitutes are demineralized bone matrix (DBM) and cellular bone allografts (CBAs). DBM is an allogeneic bone graft material produced through acid extraction of the mineral phase from cadaveric cortical bone, yielding a collagens caffold with retained matrix-bound growth factors, principally members of the bone morphogenetic protein (BMP) family, that are proposed to support osteo induction and osteo conduction at the transplant site [43]. CBAs represent a more recently developed category that incorporates viable mesenchymal stem or progenitor cells within a processed allogeneic matrix, with the intent of providing osteogenic cellular contribution in addition to the scaffold and signalling components offered by acellular preparations [131]. Both graft categories have been the subject of expanding clinical utilization and a growing body of published literature, though the evidence base for each varies in depth, design, and methodological rigor.
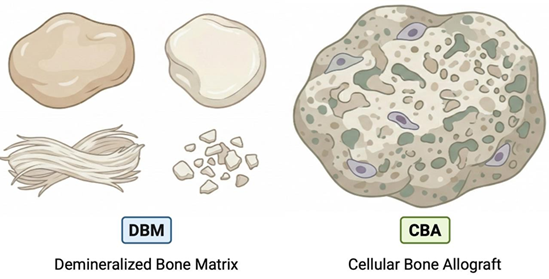
Figure 1: The image on the left depicts demineralized bone matrix (DBM), an acellular allogeneic graft material typically presented as a putty, paste, strip, fiber, or particulate-based formulation intended to provide an osteoconductive scaffold with retained matrix-associated osteoinductive potential. The image on the right depicts a cellular bone allograft (CBA), which generally consists of an allogeneic bone- derived matrix combined with viable cells, often presented in a hydrated or particulate format for surgical implantation. The figure is intended to illustrate general product configuration differences between DBM and CBA categories and is not meant to indicate comparative performance, handling superiority, or clinical efficacy.
Despite the increasing clinical adoption of both DBM and CBA products, structured comparative evaluation across the domains most relevant to clinical decision-making (biologic mechanism, clinical performance, regulatory classification, manufacturing consistency, and economic considerations) remains limited. Much of the available literature focuses on individual product evaluations or single-category reviews rather than systematic cross-category comparison. Furthermore, meaningful comparison is complicated by substantial heterogeneity within each graft class, as products marketed under the same category designation may differ meaningfully in processing methods, carrier composition, cellular content,potency specifications, and regulatory pathway.
This review presents a narrative review of DBM and CBA technologies intended for clinicians, researchers, and translational stakeholders. The analysis is organized around a translational framework that evaluates graft technologies across four interrelated domains: biologic mechanism, clinical performance within defined surgical indications, regulatory classification and evidentiary context, and economic sustainability. This multi-domain approach was adopted because no single axis of comparison adequately captures the factors that influence graft selection in practice; a product may demonstrate favorable biological properties while lacking controlled clinical evidence, or may be supported by clinical data generated under regulatory conditions that do not require demonstration of efficacy. The analysis begins with a discussion of the biological foundations of bone regeneration and their mapping to contemporary graft strategies (Section 2), followed by detailed examination of DBM technologies (Section 3) and cellular bone allografts (Section 4). A comparative clinical and translational analysis evaluates the available evidence across spinal fusion, trauma, and biologically compromised settings while addressing product heterogeneity, regulatory context, and economic considerations (Section5). Regulatory and manufacturing factors that complicate cross-product comparison (Section 6), and future directions in graft evaluation methodology are considered(Section 7). Throughout, the emphasis is on evidence-weighted interpretation rather than categorical claims of superiority, recognizing that graft selection is most appropriately guided by indication-specific evidence, patient-level risk assessment, and transparent evaluation of the evidentiary standards supporting individual products.
2. Biological Foundations of Bone Regeneration
2.1 Osteogenesis, Osteoinduction, Osteoconduction
Successful bone regeneration is commonly described through three interdependent biological principles: osteogenesis, osteoinduction, and osteoconduction [4, 5]. These principles collectively define the biologic requirements for predictable osseous healing following spinal fusion, fracture repair, or defect reconstruction, and their relative contribution varies depending on graft composition, host biology, and the local mechanical environment.
Osteogenesis refers to the direct contribution of living osteogenic cells capable of forming new bone, while osteoinduction describes the recruitment and differentiation of progenitor cells through molecular signaling, a process first characterized through the identification of bone morphogenetic proteins in demineralized bone matrix [1, 2]. Osteoconduction reflects the structural support provided by a scaffold that permits cellular attachment, migration, and vascular ingrowth into the graft site [4]. Although frequently discussed as discrete properties, these mechanisms operate simultaneously within a mechanically stabilized and biologically active environment, and the clinical performance of any graft material depends on the interplay among all three rather than on any single mechanism in isolation [5, 6].
This triad provides a useful framework for evaluating graft substitutes, as commercially available products tend to emphasize particular mechanisms while relying on host biology to supply the remainder [15, 126, 128, 129]. Some materials function primarily as structural scaffolds that support cellular infiltration and vascularization, while others are designed to deliver signaling molecules capable of recruiting and differentiating endogenous progenitor cells, and additional strategies attempt to introduce viable cellular populations intended to contribute directly to new bone formation. The relative contribution of each mechanism to clinical outcomes depends not only on graft composition but also on local biological conditions including vascular supply, inflammatory signaling, and mechanical stability at the implantation site, factors that are themselves variable across patients and surgical contexts [6, 13].
Central to osteoinduction is the activity of growth factors embedded within the extracellular matrix, which initiate signaling cascades that regulate progenitor cell recruitment, lineage commitment, and tissue remodeling during the early stages of bone repair [1, 2, 7]. The preservation and bioavailability of these factors following tissue processing is a key variable that distinguishes graft categories and contributes to differences in osteoinductive capacity across commercial preparations. Table one summarizes the three interdependent mechanisms of osteogenesis, osteoinduction, and osteoconduction whose relative contributions vary depending on graft composition, host biology, and the local mechanical environment. These mechanisms operate simultaneously rather than independently, and their interplay ultimately determines clinical outcomes.
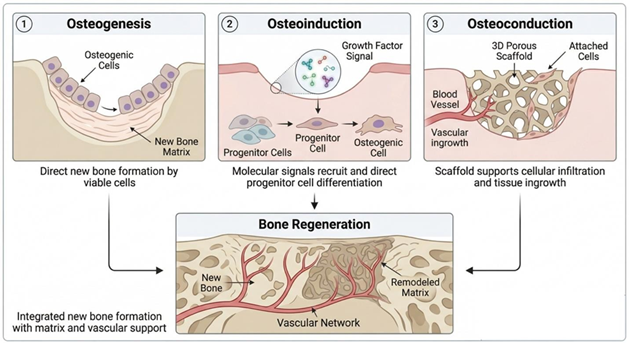
Figure 2: Schematic illustration of the three fundamental biological mechanisms involved in bone regeneration: osteogenesis, osteoinduction, and osteoconduction, and their integrated contribution to new bone formation. Panel 1 (left) depicts osteogenesis, where viable osteogenic cells directly produce new bone matrix at the defect site. Panel 2 (center) illustrates osteoinduction, in which growth factor signaling recruits progenitor cells and directs their differentiation into osteogenic cells. Panel 3 (right) demonstrates osteoconduction, where a three-dimensional porous scaffold supports cellular attachment, vascular ingrowth, and tissue infiltration. The lower panel integrates these processes, showing the formation of new bone supported by a developing vascular network and ongoing matrix remodeling.
Table 1: Interdependent Roles of Osteogenesis, Osteoinduction, and Osteoconduction in Bone Regeneration
| Feature | Osteogenesis | Osteoinduction | Osteoconduction | |
Definition | Direct formation of new bone by viable osteogenic cells | Recruitment and differentiation of progenitor cells via molecular signaling pathways | Structural support that permits cellular attachment, migration, and vascular ingrowth | |
| Primary Biological Role | Contributes living cells capable of synthesizing bone matrix | Initiates signaling cascades that drive lineage commitment and bone formation | Provides a scaffold enabling tissue infiltration and spatial guidance for new bone growth | |
Mechanism of Action | Activity of transplanted or resident osteoblasts and progenitor cells | Growth factor–mediated signaling (e.g., BMPs) that regulates progenitor cell recruitment and differentiation | Passive physical framework supporting host-driven cellular and vascular infiltration | |
| Key Biological Drivers | Viable osteogenic cells and extracellular matrix deposition | Extracellular matrix–associated growth factors (e.g.,bone morphogenetic proteins) | Scaffold architecture, porosity, and surface properties | |
| Dependence on Recipients Biology | Moderate (requires survival and integration of viable cells) | High (depends on host progenitor cell availability and responsiveness) | High (entirely dependent on host cells for bone formation) | |
| Temporal Role in Healing | Immediate contribution if viable cells are present | Early-stage activation of bone repair through cellular signaling cascades | Ongoing support throughout healing via structural guidance and vascularization | |
| Representative Graft Strategies | Cell-based therapies (CBA), autograft | Demineralized bone matrix, growth factor– enhanced grafts | Synthetic scaffolds (e.g., HA, TCP, biphasic calcium phosphate) | |
Key Limiting Factors | Cell viability, donor site morbidity, limited cell numbers | Variability in growth factor preservation and bioavailability | Lack of intrinsic biological signaling; relies on host environment | |
| Contribution to Clinical Outcomes | Direct bone formation capacity | Determines initiation and magnitude of regenerative response | Enables space maintenance and organized tissue ingrowth | |
Role in the “Triad” Framework | Cellular component of bone regeneration | Biological signaling component | Structural component of bone regeneration |
2.2 The Role of Growth Factors and Cellular Components
Osteoinductive signaling during bone repair is largely mediated by a network of growth factors that regulate progenitor cell recruitment, differentiation, and matrix production [7, 14]. Among the most extensively studied mediators are members of the bone morphogenetic protein (BMP) family, particularly BMP- 2 and BMP-7, which play central roles in initiating osteogenic differentiation of mesenchymal stem and progenitor cells through receptor-mediated signaling pathways that activate transcriptional programs responsible for osteoblast lineage commitment and extracellular matrix (ECM) deposition [3, 8, 9, 10]. DBM preparations retain many of these factors within collagen scaffolds following removal of the mineral phase, with biological activity dependent upon both preservation during processing and subsequent release kinetics at the implantation site [16, 24]. However, such preparations may also contain BMP antagonists and variable concentrations of non-osteoinductive matrix components, contributing to heterogeneity in net bioactive factor availability across commercial products [24].
Several hundreds of additional signaling molecules contribute to the broader regenerative environment. Key molecules include transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF). These growth factors coordinate angiogenesis, cellular proliferation, and early inflammatory responses that precede bone formation [11, 14, 86].
In many bone graft substitutes, these signaling molecules are not delivered as isolated recombinant proteins but are instead present within the extracellular matrix of donor-derived tissues, where they may be released gradually through scaffold degradation and cell-mediated liberation [7, 16]. This matrix- bound delivery mechanism is thought to contribute to the recruitment and differentiation of host progenitor cells, though the kinetics and dose of growth factor release vary with processing conditions, carrier composition, and scaffold architecture across commercial processing preparations.
At the same time, more recent graft technologies have attempted to supplement or augment these signaling processes by incorporating viable cellular populations [47, 50]. Such approaches seek to reduce dependence on host cell recruitment by delivering exogenous cells capable of direct osteogenic differentiation and paracrine signaling [50, 51, 132, 133]. The extent to which transplanted cells survive, engraft, and contribute functionally to repair varies by platform and remains an active area of research and clinical investigation [54, 131].
2.3 Importance of the Host Microenvironment
While growth factor signaling and cellular recruitment are central to osteoinductive activity, the regenerative potential of any graft material is constrained by the local biological environment in which it is implanted [12, 13]. Following graft placement, cells and signaling molecules must function within a dynamic microenvironment characterized by limited oxygen availability, evolving inflammatory signaling, and variable mechanical stabilization, conditions that may differ substantially across surgical sites and patient populations.
Early stages of bone repair are frequently associated with relative hypoxia and restricted nutrient diffusion until neovascularization establishes adequate perfusion [13]. During this interval, cell survival and metabolic activity depend heavily on oxygen diffusion, nutrient availability, and the rate of vascular ingrowth, all of which are influenced by scaffold porosity, architecture, and host vascular access [7, 11]. Early healing also occurs within an inflammatory milieu characterized by macrophage infiltration and cytokine signaling that can either support or impair graft integration, depending on polarization state and duration [12, 106]. Patient-specific factors also bear on this process. Genetic background and lifestyle-related influences, such as smoking status, nutrition, metabolic health, and physical activity, may substantially shape the host microenvironment and thereby affect graft integration. These variables may influence vascularity, inflammatory tone, cellular responsiveness, and remodeling capacity, further contributing to interpatient variability in regenerative outcomes [75]. These constraints highlight that graft performance is not determined solely by intrinsic composition, but by the capacity of the host microenvironment to support cellular viability, signaling persistence, and tissue remodeling [6, 13, 84].
2.4 Mapping Biological Mechanisms to Graft Categories
Contemporary graft technologies address the osteogenic triad through distinct strategic emphases, and these differences in mechanistic approach have important implications for comparative evaluation [5, 15]. Some products prioritize osteoinduction and osteoconduction through matrix-bound growth factors and scaffold architecture while relying on host biology to supply the osteogenic cellular component [16, 43]. Others incorporate viable cells with the intent of providing all three components (scaffold, signaling, and cellular osteogenesis), though the magnitude and durability of such cellular contributions in clinical environments remain under investigation, and the extent to which they translate into measurable clinical advantages over acellular alternatives has not been definitively established [47, 131]. These differing mechanistic strategies form the basis for the comparative evaluation of DBM and CBA technologies presented in the following sections.
3. Demineralized Bone Matrix Technologies
Demineralized bone matrix (DBM) represents a widely utilized class of allogeneic bone graft substitutes in orthopedic and spinal surgery [16, 43]. DBM is produced through acid extraction of cadaveric cortical bone, a process that removes the mineralized component while preserving the underlying collagen matrix and a range of matrix-bound growth factors. The resulting material is classified primarily as an osteoinductive and osteoconductive graft substrate, providing a collagen scaffold capable of supporting cellular infiltration while releasing endogenous signaling molecules that may recruit and differentiate host progenitor cells [1, 16]. Because DBM is derived from donor bone and processed through various proprietary manufacturing methods, its biological activity can vary substantially across preparations [20, 44]. Differences in donor characteristics, processing techniques, residual calcium content, carrier materials, and sterilization methods may influence growth factor preservation, scaffold architecture, and overall osteoinductive potential. DBM products are accordingly better understood as a heterogeneous category than as a single uniform graft technology [43]. Another consideration is the regulatory pathway that DBM manufacturers or processors take. While some follow a traditional medical device pathway others fall under a homologous tissue designation or Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) [101]. Medical devices are approved or cleared to make specific claims while HCT/Ps must make homologous intent usage statement(s). These regulatory classifications can dictate the labels and instructions for use (IFU). Regardless of the regulatory pathway these products are scientifically osteoinductive and osteoconductive in nature, while they may or may not be able to state this from a regulatory standpoint.
3.1 Processing and Mechanism
Demineralized bone matrix is produced through chemical extraction processes that remove the mineral phase of donor cortical bone while preserving the collagenous extracellular matrix and associated non-collagenous proteins [16]. Demineralization is most commonly achieved through exposure to dilute hydrochloric acid or similar agents, which dissolve hydroxyapatite crystals and expose matrix-bound growth factors embedded within the bone matrix [1, 16]. The extent of demineralization varies across commercial preparations, with residual calcium content ranging from negligible to substantially present [17, 19, 21]. This variation influences both growth factor bioavailability and scaffold resorption kinetics. This demineralization transforms rigid mineralized bone into a more compliant collagen scaffold in which matrix-bound signaling molecules may become more bioavailable following implantation.
The osteoinductive properties attributed to DBM arise largely from the exposure and subsequent release of growth factors retained within the extracellular matrix, particularly members of the bone morphogenetic protein (BMP) family [1, 3, 24]. Following implantation, these signaling molecules may be liberated through scaffold degradation and cell-mediated remodeling, thereby contributing to recruitment and differentiation of host progenitor populations [16, 24]. This mechanism positions DBM as a biologically active matrix proposed to promote recruitment and differentiation of host mesenchymal progenitor cells while simultaneously providing an osteoconductive framework for tissue in growth.
Commercial DBM products frequently incorporate carrier materials designed to improve handling characteristics, structural stability, and graft retention within surgical sites [18]. Common carrier materials include glycerol, hyaluronic acid, collagen gels, or other biodegradable polymers that transform particulate DBM into moldable putties, pastes, or injectable formulations [18, 43]. While carriers enhance surgical usability, they may also influence graft performance by diluting the concentration of matrix-derived growth factors or altering release kinetics within the implantation environment [24].
DBM products undergo terminal sterilization through one of several options including gamma irradiation, electron beam irradiation, or aseptic processing techniques [16, 123]. Sterilization method and dose may affect BMP retention and matrix integrity, thereby contributing to inter-product variability in biological activity [20, 123].
Donor variability and differences in manufacturing protocols produce substantial heterogeneity in osteoinductive potential across DBM products [20, 44]. Factors such as donor age, bone source, demineralization conditions, residual calcium content, sterilization parameters, and carrier composition collectively influence the preservation and bioavailability of osteoinductive proteins [19, 23]. This variability has led to considerable interest in developing standardized assays capable of assessing osteoinductive potency across preparations, although widely accepted clinical benchmarks remain limited [22, 44].

Figure 3: Schematic representation of demineralized bone matrix (DBM) processing, composition, and biological function. This schematic illustrates the production and functional mechanisms of demineralized bone matrix (DBM) used in spinal applications. Donor cortical bone undergoes acid-mediated demineralization, typically using hydrochloric acid, to remove the mineral phase while preserving the collagenous extracellular matrix and embedded growth factors. The resulting DBM is shown as a collagen-rich scaffold containing matrix-bound signaling molecules, including bone morphogenetic proteins, which contribute to osteoinductive activity.
3.2 Clinical Evidence Across Applications
Demineralized bone matrix has been evaluated across a range of orthopedic and spinal indications, most extensively as an adjunct in spinal fusion procedures and to a lesser degree in the management of fracture defects and non-union [36, 43]. In clinical practice, DBM is frequently used as a graft extender or substitute combined with autograft, allograft, or structural implants rather than as a standalone graft material [31, 43]. Reported clinical outcomes therefore reflect not only the biological activity of DBM itself but also surgical technique, fixation stability, and the characteristics of adjunct graft materials used in combination. Despite these confounding variables, a body of clinical literature has examined DBM-containing graft constructs in spinal fusion and trauma applications, providing insight into its performance across diverse surgical contexts.
3.2.1 Spinal Fusion
Spinal fusion represents the most extensively studied clinical application of DBM [29, 37]. Numerous retrospective and prospective studies have evaluated DBM as a graft extender in posterolateral lumbar fusion, cervical fusion, and interbody procedures [25, 26, 27, 28, 39]. In many of these settings, DBM is combined with local autograft or cancellous allograft to increase graft volume while attempting to augment biological activity.
Reported fusion rates in DBM-augmented constructs commonly fall within ranges comparable to traditional graft strategies when adequate mechanical stabilization is achieved [29, 37]. Several studies have described fusion outcomes in ranges overlapping those reported for autograft-containing constructs, particularly when DBM is used as an extender rather than a complete graft substitute [26, 31, 32, 35]. However, interpretation of these results is complicated by heterogeneity in DBM formulations, carrier materials, surgical techniques, and patient populations across studies [36, 38]. The literature suggests that DBM can function effectively as part of multicomponent graft constructs, but the relative contribution of DBM itself to fusion success remains difficult to isolate in many clinical series. For example, Fu et al. [26] compared DBM with autogenous iliac bone graft in multisegment posterolateral fusion and reported fusion rates of 80.8% versus 85.7%, a difference that did not reach statistical significance but demonstrated reduced donor-site morbidity in the DBM group. Buser et al. [31] conducted a systematic review of allograft and DBM in instrumented lumbar fusion and found broadly comparable radiographic fusion outcomes across graft types. Kim et al. [32] reported that DBM within a PEEK cage achieved fusion rates comparable to iliac crest autograft in anterior cervical procedures.
Table 2. Summary of Clinical Studies evaluating Demineralized Bone Matrix (DBM) in Spinal Fusion
| Author(s) & Year | Study Design | Procedure | Product | Comparator | Fusion/Union Rate (%) | Follow-Up |
| Cammisa et al. (2004) [37] | Prospective controlled | Posterolateral lumbar fusion | Grafton DBM Gel | Autograft (side- by-side) | Equivalent fusion rates at 2 years | 24 mo |
| Fu et al. (2016)[26] | Retrospective | Multi segment posterolateral lumbar fusion | DBM | Autogenous iliac bone graft | 80.8% (DBM) vs 85.7% (ICBG); p = NS | NR |
| Nam & Yi (2016)[27] | Retrospective | Posterolateral lumbar fusion | DBM | Hydroxyapatite | Compared bone union rates;outcomes reported | NR |
| Kim et al. (2017)[32] | Retrospective | ACDF (cervical) | DBM in PEEK cage | Iliac crest autograft | Comparable fusion rates | NR |
| Chin et al. (2017)[35] | Retrospective | Outpatient ACDF(cervical) | DBM in PEEK | None (single- arm) | Evaluated soft tissue swelling incidence | NR |
| Zadegan et al. (2017) [39] | Systematic review | ACDF (cervical) | DBM (multiple) | Autograft (historical) | Non-inferior to autograft across most studies | Variable |
| Buser et al. (2018) [31] | Systematic review | Instrumented lumbar fusion | Allograft vs DBM | Autograft / allograft | Similar fusion rates across graft types | Variable |
| Han et al. (2020) [29] | Meta-analysis | Posterolateral lumbar fusion | DBM + autograft | Autograft alone | No significant difference in fusion rate | Variable |
| Chang et al. (2021)[28] | Retrospective | Posterolateral lumbar fusion | Grafton vs DBX | Head-to-head DBM comparison | Similar fusion rates between products | NR |
| Eleswarapu et al. (2021)[25] | Retrospective cohort | Instrumented lumbar fusion | DBM | rhBMP-2 | 87% (DBM) vs 98.5% (rhBMP-2); lower cost/complications with DBM | NR |
3.2.2 Trauma and Non-Union Applications
Beyond spinal fusion, DBM has also been investigated in the treatment of bone defects, fracture repair, and non-union management [41, 116]. In these contexts, DBM is typically used as a filler material combined with cancellous grafts, structural allografts, or fixation constructs designed to stabilize the defect environment. Clinical reports describe bone healing in DBM- augmented constructs, although the evidence base for trauma applications remains smaller and more heterogeneous than that for spinal fusion [42, 116]. Drosos et al. [42] reviewed the comparative use of allograft and DBM in orthopedic trauma and concluded that neither material demonstrated clear superiority across all indications. Van der Stok et al. [116] examined the available clinical evidence for DBM in long bone fracture augmentation, noting acceptable healing rates but emphasizing the limited quality and heterogeneity of the existing literature.
As in spinal applications, variability in product formulation and surgical technique complicates direct comparisons across studies [42]. Differences in defect size, vascularity, and host biology may further influence clinical outcomes in trauma and non-union settings. DBM is frequently incorporated into reconstructive strategies for bone defects, yet high-quality comparative studies isolating its independent contribution remain limited [122].
3.3 Limitations and Variability
DBM exhibits several limitations that complicate both mechanistic interpretation and clinical evaluation, arising from its biological origin and manufacturing complexity [20, 44]. A central challenge is variability in osteoinductive activity across preparations. Because DBM is derived from donor bone, biological differences in donor age, skeletal site, and metabolic status may influence the concentration and activity of matrix- bound growth factors [19, 23]. These donor-dependent factors contribute to variation in osteoinductive potential prior to any processing effects.
Tissue manufacturing processes further contribute to heterogeneity across commercial DBM products [16, 20]. Differences in demineralization conditions, residual mineral content, sterilization methods, and carrier composition may influence the preservation and bioavailability of osteoinductive proteins [24, 123]. High-dose irradiation or aggressive chemical processing can reduce growth factor activity, whereas carrier materials used to improve handling characteristics may dilute the concentration of biologically active matrix components within the final allograft formulation [18, 20]. Products processed by different tissue banks or commercial entities may therefore exhibit meaningful differences in biological activity despite similar regulatory and product classification [21, 44].
Another challenge is the absence of widely accepted standardized assays for evaluating osteoinductive potency [22, 45]. Although in vivo animal assays have historically been used to assess osteoinductive activity, these models are difficult to standardize due to species variability, site-dependent response, and uncertain translation to human clinical performance [19, 22, 46]. In practice, product selection often depends on manufacturer- reported processing standards, institutional preference, or limited comparative clinical evidence. This leaves the relative biological activity of individual formulations uncertain [44].
Finally, the interpretation of clinical outcomes associated with DBM is complicated by its frequent use as a graft extender rather than a standalone graft material [31, 43]. In many surgical applications, DBM is combined with autograft, cancellous allograft, or structural implants. While this strategy can improve graft volume and handling characteristics, it also makes it difficult to isolate the independent contribution of DBM to fusion or bone healing outcomes in clinical studies [29, 36]. These limitations underscore the importance of interpreting DBM-associated clinical results within the broader context of multicomponent graft constructs and heterogeneous manufacturing practices.
4. Cellular Bone Allograft Technologies
Cellular bone allografts (CBAs) represent a more recently developed category of osteobiologic graft materials distinguished from conventional bone graft substitutes by the intentional inclusion of viable cellular populations within a processed allogeneic matrix [131]. Unlike DBM, which relies on matrix- bound growth factors and scaffold architecture to recruit and differentiate host progenitor cells, CBAs are designed to deliver mesenchymal stem cells (MSCs) or osteoprogenitor cells that may contribute directly to bone formation at the implantation site [47, 50]. This cellular augmentation strategy is predicated on the hypothesis that introducing exogenous progenitor cell populations may reduce dependence on host-derived cellular recruitment, with the intent of offering advantages in environments where endogenous progenitor cell availability is limited [55, 56]. Despite growing clinical adoption, the evidence base supporting CBAs continues to evolve, and important questions regarding cell viability, functional contribution, and comparative efficacy relative to established graft categories remain areas of active investigation [63, 77].
4.1 Biological Rationale
The theoretical foundation of CBAs rests on an attempt to address a recognized limitation of matrix-based graft technologies: reliance on the host microenvironment to supply the osteogenic cellular component necessary for de novo bone formation [5, 131]. DBM and similar preparations may provide osteoinductive signaling and osteoconductive scaffolding, but they depend on host mesenchymal progenitor cells to migrate into the graft site, receive inductive signals, and differentiate into bone-forming osteoblasts [16, 43]. In environments characterized by poor vascularity, diminished host progenitor cell availability, or significant biological compromise, such as revision surgery, radiation-affected tissue, or metabolically impaired patients, this dependence on host cell recruitment may be insufficient to achieve consistent osteogenesis [55, 85].
CBAs are designed to supplement this process by incorporating viable MSCs or osteoprogenitor cells within a structural matrix that also retains osteoconductive and potentially osteoinductive properties [47, 131]. In principle, transplanted cellular populations may contribute to bone formation through two complementary mechanisms. First, progenitor cells with osteogenic capacity may differentiate directly into osteoblasts and contribute to new bone matrix synthesis, a process analogous to the osteogenic contribution of autograft-derived cells [47, 48, 120]. Second, transplanted cells may exert paracrine effects by secreting a range of bioactive factors, including cytokines, chemokines, and extracellular vesicles, that modulate local inflammatory responses, promote angiogenesis, and enhance recruitment of endogenous progenitor populations [50, 51, 52]. The relative magnitude of these direct and paracrine contributions in the clinical implantation environment has not been fully characterized and likely varies across platforms and host conditions [54].
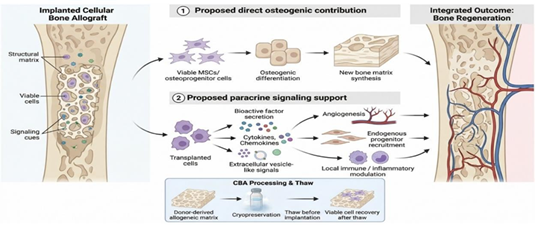
Figure 4: The combined effects of (1) the direct osteogenic contribution of live stem cells found in CBAs and (2) the additional paracrine signaling support contributing to bone regeneration.
The preservation of viable cellular constituents introduces processing complexity that distinguishes CBAs from acellular graft preparations. CBA products are typically derived from donor bone marrow, demineralized allograft matrix, or similar allogeneic substrates, and are processed using cryopreservation protocols intended to maintain cellular viability through the storage and thawing cycle [53, 54]. Cryoprotectants such as dimethyl sulfoxide (DMSO) are commonly employed to mitigate ice crystal formation and protect cell membrane integrity during freezing for allograft storage [53]. Upon thawing, the degree of cellular recovery, measured by both cell number and metabolic activity, is subject to variability related to processing parameters, storage duration, and handling at the point of implantation [54]. Commercial CBA preparations therefore aim to deliver a defined number of viable, functionally active progenitor cells, though cellular potency assays and minimum viability thresholds vary across commercial platforms [49, 131]. CBAs thus represent a strategy that attempts to reconstitute the three-component osteogenic framework, being scaffold, signaling, and cellular contribution, within a single allogeneic construct, though the net clinical benefit of the cellular component relative to acellular alternatives remains incompletely defined.
4.2 Clinical Evidence Across Applications
Clinical evaluation of CBAs has expanded considerably over the past decade, with reports spanning spinal fusion, fracture repair, and the management of bone defects and established non-union [63, 131]. The preponderance of available evidence derives from retrospective case series and single-arm prospective studies, with relatively limited representation of randomized or controlled designs [62, 77]. This evidentiary profile reflects, in part, the practical challenges associated with conducting controlled trials for graft biologics, as well as the relatively recent commercial availability of cellular allograft platforms.
Interpretation of published outcomes requires consideration of variability in study design, patient selection, adjunct fixation strategies, and endpoint definitions across reports.
In spinal fusion, several commercial CBA products have been the subject of clinical series reporting fusion rates in instrumented lumbar and cervical procedures [57, 66, 72]. Reported fusion rates in prospective cohorts have ranged from approximately 88% in cervical applications to 97–100% at 12 to 24 months of follow-up in lumbar fusion settings when outcomes are assessed per level under standardized fusion criteria [57, 58, 63, 72]. Some investigators have reported results comparable to historical benchmarks associated with iliac crest autograft, though direct comparisons are constrained by differences in patient populations, fixation constructs, and imaging-based fusion assessment methodologies [60, 65, 67, 71]. Notably, few published studies have incorporated randomized controls using DBM or other biologic comparators under equivalent surgical conditions, making definitive conclusions regarding relative efficacy difficult to establish [62, 64, 77]. The contribution of transplanted cellular elements to observed fusion rates, as distinct from scaffold-mediated and growth factor-mediated effects, has not been rigorously established through controlled mechanistic endpoints.
In trauma and non-union applications, the clinical literature for CBAs remains smaller and more heterogeneous than that available for spinal indications [70]. Some retrospective series describe union rates when cellular grafts are used as adjuncts in fracture defect repair or revision non-union management, including in settings where prior grafting attempts had been unsuccessful [59, 61, 68, 130]. These reports have contributed to interest in CBAs in biologically challenging environments where the potential benefit of exogenous progenitor cell delivery may be most clinically relevant. However, as with the spinal fusion data, these reports are predominantly retrospective, involve variable patient populations, and lack standardized comparator arms [77]. The absence of prospective head-to-head comparisons with established DBM products under controlled conditions and defined endpoints makes it difficult to isolate the incremental contribution of the cellular component to observed healing outcomes [78].
Table 3. Summary of Clinical Studies Evaluating Cellular Bone Allografts (CBAs)
| Author(s) & Year | Study Design | Procedure | Product | Comparator | Fusion/Union Rate (%) | Follow- Up |
| Kerr et al. (2011)[66] | Retrospective | Lumbar interbody fusion | Osteocel | rhBMP-2 | CBA suggested as viable alternative to rhBMP-2 | NR |
| Hollawell (2012) [68] | Retrospective | Hindfoot and ankle fusion | Trinity Evolution | Autograft (historical) | Favorable fusion outcomes reported | NR |
| Tohmeh et al. (2012)[65] | Prospective | XLIF (lateral lumbar interbody) | Osteocel Plus | None (single- arm) | High fusion rates at 12 months | 12 mo |
| Eastlack et al. (2014)[71] | Prospective multicenter | ACDF (cervical) | Osteocel Plus | None (single- arm) | High radiographic and clinical fusion rates | 12 mo |
| Jones et al.(2015)[70] | Prospective multicenter | Foot and ankle arthrodesis | Trinity ELITE | None (single- arm) | Favorable fusion outcomes | 12 mo |
| Vanichkachorn et al. (2016) [72] | Prospective | Single-level ACDF (cervical) | Trinity Evolution | Historical controls | 93.5% fusion at 12 months | 12 mo |
| Hsieh et al. (2019)[63] | Systematic review | Spinal fusion (multiple) | Multiple CBA products | Various comparators | Overall favorable fusion rates across studies | Variable |
| Elgafy et al. (2021)[59] | Retrospective | Lumbar spine fusion | V-CBA | None (single- arm) | Fusion outcomes reported with lineage-committed cells | NR |
| Saitta et al. (2021)[67] | Prospective | Multilevel ACDF (cervical) | ViviGen CBA | None (single- arm) | High 1-year fusion rates and favorable PROs | 12 mo |
| Goldman et al. (2024)[60] | Prospective | ACDF (cervical) | ACBM | None (single- arm) | Favorable radiographic and clinical outcomes | NR |
| Lansford et al. (2024)[57] | Prospective | Lumbar spinal fusion | Trinity Elite | None (single- arm) | High fusion rates irrespective of surgical risk factors | 12 mo |
| Russo et al. (2024)[58] | Prospective | Lumbar spinal fusion | Trinity Elite | None (single- arm) | Maintained high fusion rates despite risk factors for non-union | 12 mo |
4.3 Limitations and Open Questions
Despite the biological rationale supporting CBA technologies and the outcomes reported in available clinical series, several important uncertainties remain unresolved and merit careful consideration in the evaluation of this graft category [77]. A central question concerns the extent to which transplanted cells survive and maintain functional activity following implantation in human bone defect or fusion environments [76]. The early post- implantation period is characterized by relative hypoxia, limited nutrient availability, and an evolving inflammatory milieu that may adversely affect cellular viability before adequate neovascularization is established [12, 13]. Direct evidence of long-term cellular engraftment and osteogenic contribution at human implantation sites is limited, and the degree to which initial cellular content translates into sustained functional activity in vivo has not been comprehensively characterized across commercial platforms [54, 131].
Cryopreservation and thawing protocols introduce additional sources of variability that may affect product consistency and delivered cellular potency [53, 54]. Differences in cryoprotectant formulations, freeze-thaw cycle parameters, post-thaw handling requirements, and storage duration can influence cellular recovery and metabolic activity following reconstitution. Because many CBA products require cold-chain logistics and specific preparation protocols at the point of care, departures from recommended handling procedures may reduce viable cell content prior to implantation, with potentially meaningful consequences for biological activity for the recipient [54]. Standardized viability thresholds and potency release criteria vary across commercial products and have not been harmonized through broadly accepted industry benchmarks, limiting the comparability of cellular specifications across platforms [49, 77].
Regulatory classification of CBA products adds further complexity to comparative evaluation [101, 131]. Depending on processing methods and claimed indications, CBA products may be classified as HCT/Ps under Section 361 of the Public Health Service Act, subject to donor screening and tissue banking regulations but not required to demonstrate clinical efficacy through premarket review, or as biologics under Section 351, which carries a substantially more rigorous evidentiary standard [101, 146]. This regulatory heterogeneity means that products within the same clinical category may not be held to equivalent evidentiary requirements, complicating cross-product comparison and the interpretation of published outcome data based on the regulations and what specific claim indications can be made or stated. Heterogeneity across commercial platforms with respect to cellular composition, matrix source, processing methods, and potency specifications further limits the generalizability of findings from individual product studies to the broader CBA category [63, 131]. Results reported for one commercial preparation cannot be assumed to reflect the performance of other products, even when surface-level cellular viability metrics appear similar [78]. The difficulty of isolating the independent contribution of transplanted cells from matrix- mediated osteoinduction and scaffold effects further complicates attribution: clinical outcomes associated with CBA constructs cannot be credited to the cellular component alone without controlled mechanistic data [69, 77].
Long-term controlled trials evaluating CBA performance against established graft standards across defined surgical indications remain limited, and the durability of reported outcomes over extended follow-up periods has not been uniformly characterized [73, 74]. These uncertainties collectively underscore the importance of structured, comparative evaluation that accounts for product-level heterogeneity, regulatory context, and mechanistic distinction. A systematic framework for cross- category comparison, integrating biological mechanism, clinical performance data, regulatory pathway, and economic considerations, is developed in Section 5 of this review.
5. Comparative Clinical and Translational Analysis
Clinical decision-making in bone grafting ultimately depends not on theoretical osteogenic potential alone, but on reproducible patient outcomes across defined surgical contexts, interpreted through the lens of evidence quality, regulatory pathway, and economic sustainability. While both demineralized bone matrix (DBM) and cellular bone allografts (CBAs) are intended to support bone regeneration, their comparative efficacy is highly context-dependent and influenced by surgical indication, graft formulation, host biology, and the evidentiary standards under which clinical data have been generated [81]. Although CBAs are designed to augment osteogenesis through the inclusion of viable mesenchymal stem or progenitor cells, DBM products have demonstrated fusion and defect-healing rates that are broadly comparable to those reported for other graft categories across multiple orthopedic and spinal applications [29, 82]. An evaluation of the available clinical literature suggests that, despite mechanistic distinctions, categorical superiority between these graft categories has not been established and appears to vary by indication, study design, and product selection [136].
5.1 Comparative Evidence in Spinal Fusion
Spinal fusion represents the most extensively studied clinical indication for both DBM and CBAs, and the setting in which comparative evaluation is most feasible. Systematic reviews of graft alternatives in lumbar fusion have reported that DBM and structural allograft achieve broadly comparable fusion rates in instrumented procedures, with differences emerging primarily in non-instrumented settings where osteoinductive capacity may assume greater relative importance [79, 31]. Reported fusion rates for commercially available DBM products frequently range between 80 and 100 percent, depending on formulation, fixation strategy, and patient-specific risk factors, while CBA products have reported rates of approximately 88 percent in cervical applications and 97 to 100 percent in lumbar settings [29, 63]. These rates were assessed at 12 to 24 months postoperatively, though follow-up duration varied across studies. In anterior cervical discectomy and fusion, comparative cohort data suggest that cellular and noncellular allografts achieve similar radiographic fusion rates and clinical outcomes when used with contemporary interbody devices [139, 140], a finding consistent with broader systematic reviews indicating substantial overlap in outcome ranges across graft categories [83].
Table 4. Comparative Clinical Evidence in Spinal Fusion: DBM vs CBA
| Graft Category | Author(s) &Year | Study Design | Procedure | Product | Comparator | Fusion/Union Rate (%) | Follow-Up |
| DBM | Cammisa et al. (2004)[37] | Prospective controlled | Posterolateral lumbar fusion | Grafton DBM Gel | Autograft (side-by-side) | Equivalent fusion rates at 2 years | 24 mo |
| DBM | Fu et al. (2016) [26] | Retrospective | Multi segment PLF | DBM | ICBG | 80.8% vs 85.7%; p = NS | NR |
| DBM | Kim et al. (2017)[32] | Retrospective | ACDF (cervical) | DBM in PEEK cage | ICBG | Comparable fusion rates | NR |
| DBM | Zadegan et al. (2017) [39] | Systematic review | ACDF (cervical) | DBM (multiple) | Autograft (historical) | Non-inferior to autograft | Variable |
| DBM | Buser et al. (2018) [31] | Systematic review | Instrumented lumbar fusion | Allograft vs DBM | Autograft / allograft | Similar fusion rates | Variable |
| DBM | Han et al. (2020)[29] | Meta- analysis | PLF (lumbar) | DBM + autograft | Autograft alone | No significant difference | Variable |
| DBM | Eleswarapu et al. (2021) [25] | Retrospective cohort | Instrumented lumbar fusion | DBM | rhBMP-2 | 87% vs 98.5%; lower cost with DBM | NR |
| CBA | Kerr et al. (2011)[66] | Retrospective | Lumbar interbody fusion | Osteocel | rhBMP-2 | CBA as viable alternative | NR |
| CBA | Tohmeh et al. (2012) [65] | Prospective | XLIF (lumbar) | Osteocel Plus | None (single-arm) | High fusion rates | 12 mo |
| CBA | Eastlack et al. (2014) [71] | Prospective multicenter | ACDF (cervical) | Osteocel Plus | None (single-arm) | High fusion rates | 12 mo |
| CBA | Vanichkachorn et al. (2016)[72] | Prospective | Single-level ACDF | Trinity Evolution | Historical controls | 93.5% at 12 months | 12 mo |
| CBA | Hsieh et al. (2019) [63] | Systematic review | Spinal fusion(multiple) | Multiple CBA products | Various | Favorable fusion rates | Variable |
| CBA | Saitta et al. (2021) [67] | Prospective | Multilevel ACDF | ViviGen CBA | None (single-arm) | High 1-year fusion rates | 12 mo |
| CBA | Lansford et al. (2024)[57] | Prospective | Lumbar spinal fusion | Trinity Elite | None (single-arm) | High rates irrespective of risk factors | 12 mo |
| CBA | Russo et al. (2024) [58] | Prospective | Lumbar spinal fusion | Trinity Elite | None (single-arm) | Maintained high rates despite risk factors | 12 mo |
| Cross-Cat. | Fischer et al. (2013) [136] | Systematic review | Lumbar/cervical fusion | Multiple graft types | Cross-category | High heterogeneity; no definitive recommendation | Variable |
| Cross-Cat. | Tuchman et al. (2016) [79] | Systematic review | Lumbar spinal fusion | ICBG vs local/allograft | Cross-category | Comparable fusion; allograft less morbidity | Variable |
| Cross-Cat. | Tuchman et al. (2017) [140] | Systematic review | Cervical spinal fusion | Autograft vs allograft | Cross-category | Similar fusion rates | Variable |
| Cross-Cat. | Li et al. (2022) [83] | SR / Meta- analysis | Spine surgery (general) | Multiple graft categories | Cross-category | 64 articles; fusion rates across graft types | Variable |
| Cross-Cat. | Zakko et al. (2023) [139] | Comparative cohort | ACDF (cervical) | Cellular vs non cellular | Head-to-head | Similar fusion and clinical outcomes | NR |
Despite these reports, interpretation requires attention to study design limitations. The majority of available data for both graft categories, DBM and CBA, derive from retrospective series, single-arm prospective cohorts, or studies with industry sponsorship. Few rigorously designed head-to-head randomized controlled, level one trials directly compare DBM and CBA products under equivalent surgical conditions [81]. Meta-analytic efforts have been constrained by heterogeneity in endpoint definitions, imaging-based fusion assessment criteria, and inconsistent adjustment for confounding variables including fixation construct, smoking status, and metabolic comorbidities [118, 119, 136]. The relative contribution of transplanted cellular elements to observed fusion outcomes, as distinct from scaffold- mediated and growth factor–mediated effects, has not been isolated through controlled mechanistic endpoints in any published comparative trial [138].
An additional consideration in interpreting these data is the asymmetry in evidence maturity between the two graft categories. The clinical literature supporting DBM spans more than two decades and includes multiple meta-analyses, systematic reviews, and prospective controlled comparisons, whereas the CBA evidence base is concentrated within the past ten to twelve years and remains predominantly composed of single-arm prospective cohorts and retrospective case series [63, 77, 131]. This difference in evidentiary depth does not inherently favor one category over the other, but it does mean that the confidence intervals around DBM performance estimates are generally narrower, and that the consistency of reported outcomes has been tested across a broader range of surgical contexts, patient populations, and product formulations. Comparisons drawn from overlapping fusion rate ranges should therefore be interpreted with attention to the volume, design, and independence of the studies generating those estimates.
5.2 Trauma, Non-Union, and Biologically Compromised Settings
Beyond spinal fusion, both DBM and CBAs are utilized in the management of traumatic bone defects, established non-unions, and reconstructive procedures in which host osteogenic capacity may be compromised. In these settings, graft selection is influenced by defect geometry, vascularity, mechanical stability, and the availability of endogenous progenitor cell populations. Evidence reviews of bone graft substitutes in orthopedic trauma have noted acceptable healing rates with multiple graft categories, while emphasizing that surgical technique, mechanical fixation, and host biology often exert greater influence on union outcomes than graft material selection alone [88, 122]. Some retrospective series describe union rates when cellular grafts are used as adjuncts in revision non-union cases, though these reports involve heterogeneous patient populations and lack standardized comparator arms [70, 141].
The rationale for CBAs in biologically compromised environments, including patients with smoking history, diabetes, osteoporosis, or prior radiation exposure, rests on the premise that exogenous progenitor cell delivery may compensate for diminished host cell recruitment [40, 89, 90]. While this rationale is biologically plausible, definitive clinical evidence establishing superior outcomes for CBAs over comparable DBM preparations in these specific populations remains limited [91, 92]. The absence of prospective comparative data stratified by host risk factors represents a notable gap in the current evidence base and an area where future investigation may clarify the clinical circumstances under which cellular augmentation confers meaningful incremental benefit.
It bears noting that clinical adoption of CBAs in biologically compromised settings has, in some respects, outpaced the controlled evidence supporting their preferential use in those populations. The biological rationale for delivering exogenous progenitor cells to environments with diminished host recruitment capacity is sound in principle, but the published literature does not yet include prospective trials that stratify CBA and DBM outcomes by specific host risk factors under equivalent surgical conditions. The gap between the plausibility of the mechanism and the rigor of the supporting clinical data is a recurring theme in this field, and it underscores the importance of distinguishing between what a graft technology is designed to do and what it has been demonstrated to do in controlled settings.
5.3 Product Heterogeneity and Evidence Generalizability
A persistent challenge in comparative graft evaluation is the substantial heterogeneity that exists both within and between graft categories. Among DBM products, donor-to-donor variability in retained bone morphogenetic protein content, differences in demineralization protocols, and the influence of carrier formulations on handling and resorption characteristics contribute to meaningful inter-product performance differences that are not captured by class-level comparisons [44]. Similarly, CBA products vary considerably in cellular composition, matrix source, cryopreservation protocols, viability thresholds, and potency release criteria, limiting the generalizability of findings from individual product studies to the broader category [77, 131]. Results reported for one commercial preparation cannot be assumed to reflect the performance of other products within the same class, even when surface-level specifications appear similar [143]. This product-level heterogeneity complicates the interpretation of systematic reviews and meta-analyses that aggregate data across platforms, as class-level effect estimates may obscure clinically relevant differences between specific formulations [136, 127, 144]. Comparative evaluation would benefit from product-specific outcome registries and standardized potency assays that permit more precise characterization of the relationship between graft composition and clinical performance [145]. Table five demonstrates a non-comprehensive list of DBM and CBA known products within the market.
Table 5. Representative Commercial DBM and CBA Products: Comparative Translational Features
| Product | Class | Processing / Preservation Features | Representative Clinical Use(s) | Major Comparative Caveat | Ref(s) |
| Grafton DBM | DBM | Aseptic processing; low-dose gamma irradiation (1.0-1.5 Mrad) | Posterolateral lumbar fusion (graft extender); bone void filling in spine and orthopedic procedures | Glycerol carrier may dilute effective DBM concentration; donor variability in growth factor content affects lot-to- lot performance | [37] |
| DBX | DBM | Demineralization with lot-level osteoinductive potency testing (validated ectopic bone assay) | Bone void filling; graft extender in spinal and trauma applications | Carrier composition differs from glycerol-based products; limited published comparative data relative to other DBM platforms | [16] |
| Accell Evo3 | DBM | Aseptic processing from AATB-accredited tissue bank | Posterolateral lumbar fusion; bone void filling | Reverse-phase carrier mechanism differs from standard glycerol/sodium hyaluronate carriers used in other DBM products | [184], [193]† |
| Progenix Plus | DBM | Demineralization with two- stage osteoinductive testing | Spinal fusion; bone defect applications | Bovine collagen carrier introduces xenogeneic component absent from other listed DBM products | [36], [188]† |
| InterGro DBM | DBM | Demineralization optimized for osteoinductivity; lecithin carrier | Bone void filling in spine and orthopedic applications | Lecithin carrier is compositionally distinct from glycerol-based DBM products | [19], [191]† |
| Optium DBM | DBM | PAD technology for controlled residual calcium levels | Spinal fusion; orthopedic and neurosurgery applications | Clinical literature frequently pools glycerol-carrier DBMs without product-level differentiation | [38] |
| Magnifuse | DBM | Aseptic processing with optional low-dose gamma irradiation | Posterolateral spine fusion; bone void filling | Multi-component design and containment mesh distinguish it from single-component DBM formulations | [184], [189]† |
| Staysis DBM | DBM | 100% allograft bone with no synthetic carriers; room temperature storage; 2-year shelf life; available as putty and crunch (putty + cancellous chips)forms | Bone void filling; repair, replacement, or reconstruction of bony defects of the skeletal system | Recently introduced carrier-free DBM platform; independent peer-reviewed clinical outcome data not yet available at time of publication; author disclosure applies | [34]† |
| AlloFuse DBM | DBM | Validated Sterilizer disinfection (SAL 10⁻⁶). AATB-accredited processing | Spinal fusion; extremity and pelvis applications | Multiple formulation variants span acellular and cellular categories, complicating class-level comparison | [16], [190]† |
| Osteocel Plus / Osteocel Pro | CBA | Cryopreserved at −80°C. Donor tissue recovered within 24h of death | ACDF (87–92% fusion reported); TLIF(91% at 12 mo) | Evidence largely retrospective and single-arm. Section 361 HCT/P pathway does not require premarket efficacy | [169], [67] |
| Trinity ELITE | CBA | Cryopreserved in liquid nitrogen at −185°C. Strict donor selection with viability testing | ACDF (93.5% SL, 97.4% ML high-risk fusion); PLF (97– 100% at 12–24 mo) | Among the most published CBA platforms, but evidence base is predominantly prospective single-arm | [74], [186] |
| ViviGen | CBA | Cryopreserved viable cells. Proprietary preservation protocol | Spinal fusion; foot/ankle arthrodesis; trauma/extremity fusion | Manufacturer’s distinction between “lineage-committed bone-forming cells” and MSCs lacks independent verification | [185], [70] |
| Cellentra VCBM | CBA | Cryopreservation method; specific parameters not publicly disclosed | Posterolateral spinal fusion. Prospective ACDF trial | Publicly available product specifications are less detailed than those for competing CBA products | [187], [192]† |
| Map3 | CBA | Cryogenic preservation of MAPC cells. Available in putty and strip forms | ALIF (fusion rates comparable to rhBMP-2 in retrospective data) | MAPC cell type is proprietary and distinct from MSCs; limited independent peer-reviewed clinical data | [137] |
| Bio4 | CBA | BioSmart proprietary cryopreservation technology (no DMSO) | Bone void filling; foot/ankle bone regeneration; trauma | Rebranded from OvationOS following 2019 Stryker acquisition; clinical evidence is limited relative to longer- established CBA platforms | [142]† |
† Denotes non-peer-reviewed source (FDA 510(k) summary,ClinicalTrials.gov registration, or manufacturer product documentation). These sources are included in the manuscript bibliography and are flagged for transparency regarding evidence type.
5.4 Regulatory Classification and Evidentiary Context
The regulatory framework governing DBM and CBA products adds further complexity to comparative evaluation. Depending on product composition, processing, and intended use, DBM and CBA products may be regulated either as human cells, tissues, and cellular and tissue-based products (HCT/Ps) under Section 361 of the United States Public Health Service Act or, alternatively, as biological products under Section 351, which carries substantially more rigorous evidentiary requirements [101, 146]. Products marketed as HCT/Ps are subject to donor screening and tissue banking requirements but are not required to undergo premarket review for clinical efficacy. In addition, DBM and CBA products regulated as HCT/Ps are not permitted to include mechanism-based claims in their labeling. Nevertheless, scientific evidence suggests that these materials may exhibit osteoinductive and osteoconductive properties based on their composition and retained growth factor content. As a result, although these biological functions are commonly described in the scientific literature, commercially marketed HCT/P products are limited to homologous use statements and do not explicitly reference such mechanisms in their labeling. This regulatory heterogeneity carries a practical consequence that is often underappreciated in comparative evaluation: products distributed under the HCT/P framework can reach widespread clinical adoption without generating controlled efficacy data of the type that would be required under a biologics or device pathway. As a result, the evidence supporting individual products within the same clinical category may differ not only in quantity and quality, but in kind, with some products supported primarily by post-market observational experience and others by premarket controlled investigation. This asymmetry complicates cross-product comparison and means that similar clinical adoption levels should not be interpreted as reflecting equivalent evidentiary support. The regulatory pathway through which a product is marketed has important implications for the nature of the evidence available in support of its use. Products distributed under tissue banking frameworks are commonly supported by donor screening, processing controls, preclinical and bench characterization, and post-market clinical experience, while biologic products are generally supported by additional controlled clinical investigation requirements [99, 147]. As such, consideration of regulatory pathway can provide helpful context when interpreting the rigor, scope, and type of evidence reported across published clinical studies [98].
5.5 Economic Considerations
Economic analysis represents an important but underdeveloped component of comparative graft evaluation. Within commercially available alternatives, DBM products are generally positioned at a lower acquisition cost compared with CBAs, reflecting differences in processing complexity, cold-chain logistics, and the inclusion of viable cellular components. Health economic modeling in spinal fusion has suggested that cost-effectiveness depends not solely on upfront material costs but on downstream outcomes including revision surgery rates, hospital length of stay, and return-to-function timelines [93, 94, 95]. In scenarios where reported clinical outcomes overlap substantially between graft categories, the higher acquisition cost of CBAs may warrant scrutiny absent clear evidence of superior healing rates or reduced complications [96, 148]. Conversely, the lower acquisition cost of DBM does not inherently establish cost- effectiveness when well-documented lot-to-lot variability in osteoinductive potency is considered, as inconsistent biological activity may contribute to unpredictable clinical performance and associated downstream costs [44, 96].
To date, no high-quality cost-effectiveness analysis has directly compared commercially available DBM and CBA products under equivalent surgical conditions and standardized clinical endpoints [149, 150]. Developing such analyses will require integration of product-specific clinical outcome data, procedure- level cost accounting, and long-term follow-up sufficient to capture differences in revision rates and functional recovery [96]. Until such data are available, cost-benefit considerations remain closely tied to the strength and specificity of supporting clinical evidence.
5.6 Toward a Structured Comparative Framework
The preceding analysis underscores the difficulty of rendering categorical judgments regarding graft superiority across diverse clinical contexts. Rather than framing the comparison as a binary question of which graft category is superior, a more productive approach involves structured, multi-domain evaluation that accounts for biological mechanism, clinical performance within specific indications, regulatory pathway, product-level heterogeneity, and economic sustainability. Such a framework acknowledges that graft selection is optimally guided by indication-specific evidence, patient-level risk assessment, and transparent evaluation of the evidentiary standards supporting individual products [81, 103]. Consensus recommendations for orthobiologic evaluation have emphasized the need for standardized product characterization, minimum clinical trial design standards, and patient registries that can support comparative effectiveness research across graft platforms [103, 151]. Until rigorous head-to-head comparative trials with standardized endpoints are conducted, definitive conclusions regarding the relative merits of DBM and CBA technologies across clinical applications will remain incompletely defined.
6. Regulatory and Manufacturing Considerations
The comparative evaluation of demineralized bone matrix (DBM) and cellular bone allografts (CBAs) is complicated not only by differences in biological mechanism, scientific study support and clinical evidence, as discussed in preceding sections, but also by the regulatory and manufacturing frameworks under which individual products are developed, classified, and brought to market. These frameworks determine the regulatory standards applied to each product, the scope of permissible clinical claims, and the degree of manufacturing consistency required across production lots. Because regulatory classification and manufacturing processes differ both between and within graft categories, an understanding of these factors is essential for interpreting the published comparative literature and assessing the generalizability of product-specific clinical findings.
6.1 Regulatory Classification Framework
In the United States, non synthetic bone graft products may be regulated through several distinct pathways depending on their composition, processing, and intended use. Human cells, tissues, and cellular and tissue-based products (HCT/Ps) that meet the criteria for minimal manipulation and homologous use under Section 361 of the Public Health Service Act are subject to donor screening, tissue banking, and establishment registration requirements but are not required to demonstrate clinical efficacy through premarket review [101, 146]. Products that do not satisfy these criteria, or that incorporate more-than- minimally-manipulated cellular components, may be classified as biological products under Section 351, which requires submission of a biologics license application (BLA) supported by clinical evidence of safety and efficacy [99]. Certain DBM formulations may also follow traditional medical device pathways, including 510(k) clearance, depending on their composition and intended use claims [105, 157].
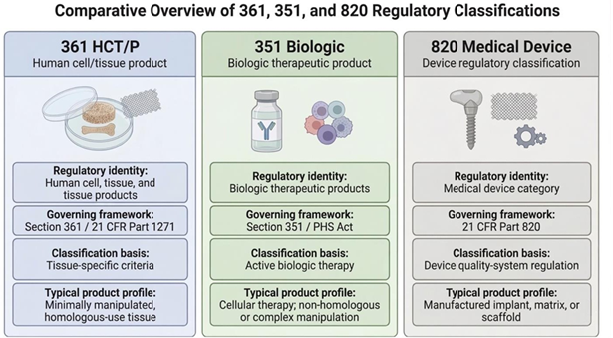
Figure 5: Differences in regulatory classifications between Section 361 HCT/Ps (left),Section 351 Biologics (middle), and Section 820 Medical Devices (right).
This regulatory heterogeneity means that products marketed for overlapping clinical indications may differ substantially in evidentiary requirements for market access. Products classified under tissue banking frameworks may enter clinical use supported by donor safety screening, manufacturing characterization and post-market surveillance, whereas products regulated as biologics or devices must meet different structured premarket standards [100]. These pathway-dependent differences have direct implications for interpreting published clinical data, as the regulatory context under which a product was commercialized provides important information about the independence and design of its supporting evidence base [147, 157].
6.2 Minimal Manipulation and Homologous Use
The regulatory criteria of minimal manipulation and homologous use are central to the classification of allograft-based bone products and merit careful definition. For structural tissues such as bone, minimal manipulation generally requires that processing does not alter the original relevant characteristics of the tissue relating to its utility for reconstruction, repair, or replacement [101]. Homologous use requires that the product performs the same basic function or functions in the recipient as in the donor. When both criteria are met, along with additional requirements regarding combinational products and systemic effects, the product may qualify for regulation solely as an HCT/P under Section 361 without premarket clinical review. The application of these criteria to bone graft products is not always straightforward. Demineralization, for example, removes the mineral phase of bone and alters its structural properties, and the regulatory classification of demineralized products depends on how the resulting material is positioned relative to the original tissue’s relevant characteristics [16]. For cellular allografts, the determination of whether cryopreservation, cell selection, or other processing steps constitute more than minimal manipulation depends on sponsor-specific details regarding processing protocols and intended use [99]. Products within the same general category may therefore receive different regulatory classifications based on their specific manufacturing details, claims, or intended uses, and generalizations about the regulatory status of an entire graft class should be made cautiously.
6.3 Manufacturing and Processing Variability
Manufacturing variability represents a significant but underappreciated source of heterogeneity across commercially available bone graft products. For DBM, inter-product variability arises from multiple sources including donor characteristics, bone procurement and storage practices, demineralization protocols, residual calcium content, terminal sterilization methods and dose, and the composition of carrier materials used to achieve desired handling properties [16, 18]. Gamma irradiation, widely employed for terminal sterilization, has been shown to affect osteoinductive activity in a dose-dependent and moisture-dependent manner, with potential reduction in retained growth factor bioactivity at higher radiation exposures [152, 153]. Alternative terminal sterilization approaches include electron beam (e-beam) irradiation, which delivers comparable sterilizing doses over shorter exposure times and may reduce cumulative protein degradation relative to gamma irradiation at equivalent doses, and chemical sterilization agents such as ethylene oxide or peracetic acid, which operate through distinct mechanisms but may introduce residual chemical effects on matrix composition [152, 153]. Aseptic processing, in which sterility is maintained through controlled environmental and procedural conditions without a terminal sterilization step, avoids radiation-induced or chemical damage to matrix proteins but depends on rigorous process validation and environmental controls to ensure microbiological safety. Studies examining lot- to-lot variability within individual DBM products have demonstrated substantial differences in bone morphogenetic protein content and in vivo osteoinductive performance across production lots from the same manufacturer, underscoring that even within a single commercial product, consistency cannot be assumed [44].
For CBAs, manufacturing variability extends to additional parameters including cellular composition, tissue sourcing, cryopreservation protocols, cryoprotectant selection and concentration, post-thaw handling requirements, viability thresholds, and potency release criteria [154, 155]. Systematic reviews of cryopreservation effects on mesenchymal stromal cells have reported that while core phenotypic markers and differentiation capacity are generally preserved, effects on viability, paracrine function, and immunomodulatory properties may vary depending on freezing methodology and post-thaw assessment timing [154]. Potency assays used to characterize cellular activity prior to release differ across commercial platforms, and no broadly accepted standardized potency benchmark exists for cellular bone allografts, limiting the comparability of cellular specifications across products [155, 156].
6.4 Implications for Evidence Interpretation
The regulatory and manufacturing considerations described above have direct consequences for evidence interpretation. When products classified under different regulatory pathways are compared on the basis of published clinical series, differences in study design and evidentiary independence may reflect not only investigator choices but also the regulatory expectations applied to each product category. Products brought to market through HCT/Ps pathways may generate evidence primarily through post-market observational data, while those regulated as biologics may produce controlled trial data of higher methodological rigor, though with narrower indications.
Manufacturing heterogeneity further limits the generalizability of product-specific findings to broader graft categories. Class-level conclusions derived from aggregating data across products with different processing methods, carrier formulations, sterilization parameters, or cellular potency specifications may obscure clinically meaningful differences between individual formulations [22, 136]. This consideration is particularly relevant when interpreting systematic reviews and meta-analyses that pool data across heterogeneous products, as the resulting effect estimates may not reliably characterize the performance of any individual product within the class.
These regulatory and manufacturing considerations, viewed as a whole, reinforce the need for product-level rather than class-level evaluation when assessing the comparative merits of DBM and CBA technologies. Readers of the comparative literature should consider regulatory pathway, manufacturing specifications, and evidence design alongside clinical outcomes when evaluating the strength and applicability of published data to specific clinical contexts.
7. Future Directions in Bone Graft Evaluation
While it is evident that bone allografts demonstrate degrees of clinical efficacy the preceding sections have identified several recurring limitations in the comparative evaluation of demineralized bone matrix and cellular bone allografts, including heterogeneity in manufacturing processes, variability in regulatory classification, reliance on retrospective and single-arm study designs, and the absence of standardized metrics for assessing biological potency across products and platforms. Addressing these limitations will require coordinated advances across multiple domains, including assay development, clinical trial design, biomarker discovery, and data infrastructure. While progress in each of these areas is ongoing, the translation of emerging methods into clinical practice remains at an early stage, and the extent to which any single advance will fundamentally alter the comparative evidence landscape is not yet clear.
7.1 Standardized Potency and Characterization Assays
A central challenge in bone graft evaluation is the absence of broadly accepted assays capable of quantifying the biological potency of graft products in a manner that permits meaningful cross-product and cross-category comparison. For DBM, osteoinductive activity has historically been assessed through in vivo ectopic bone formation models, most commonly the athymic rat or nude mouse assay [19]. However, these models are resource-intensive, difficult to standardize across laboratories, and subject to species-dependent variability that limits translation to human clinical performance [134]. In vitro approaches using alkaline phosphatase induction in responsive cell lines have been proposed as alternatives, offering more rapid and quantitative readouts, though the correlation between in vitro potency and clinical osteoinductive activity remains incompletely validated [22]. For cellular bone allografts, potency characterization is further complicated by the need to assess not only matrix-mediated osteoinduction but also the viability and functional capacity of transplanted cellular populations. Potency assays for mesenchymal stromal cell products have been the subject of increasing attention, with combinatorial assay matrices and multiplex functional readouts proposed as strategies for capturing the multidimensional nature of cellular activity [155, 156]. Nevertheless, no single standardized potency benchmark has been adopted across the cellular allograft field, and the relationship between in vitro potency metrics and clinical healing outcomes has not been prospectively established. Development of validated, clinically predictive potency assays represents a prerequisite for meaningful comparative evaluation, as without such benchmarks, comparisons between products or categories remain limited to clinical endpoint data that may be confounded by differences in manufacturing, regulatory pathway, and study design.
7.2 Comparative Clinical Trial Design
The paucity of rigorously designed head-to-head comparative trials remains one of the most significant barriers to evidence- based graft selection. As noted throughout this review, the majority of published clinical evidence for both DBM and cellular bone allografts derives from retrospective series, single-arm cohort studies, or studies comparing a single product to autograft rather than to other commercial graft alternatives. The practical barriers to conducting randomized controlled trials for bone graft biologics are well recognized and include patient enrollment challenges in the setting of surgeon preference and product availability, difficulty in blinding graft identity, variability in fixation constructs and surgical technique across sites, and the relatively long follow-up periods required to capture meaningful differences in fusion or healing endpoints [80, 81].
Pragmatic trial designs, adaptive randomization strategies, and registry-embedded randomized trials have been proposed as approaches that may overcome some of these barriers while preserving methodological rigor [151]. Such frameworks could facilitate direct comparisons between graft categories or between individual products under conditions more reflective of routine clinical practice than traditional efficacy trials. However, the feasibility and adoption of these designs within the orthobiologic field remain limited, and their successful implementation will depend on collaborative infrastructure involving surgeons, regulatory authorities, and industry stakeholders willing to support comparisons that may not favor any single commercial product but rather the field as a whole.
7.3 Biomarker-Guided Graft Selection
The concept of biomarker-guided or patient-specific graft selection represents an emerging area of investigation with the potential to move graft decision-making beyond categorical assumptions toward individualized strategies informed by host biology. Clinical experience and observational data suggest that patient-specific factors including smoking status, metabolic comorbidities, vascularity, and prior surgical history influence healing outcomes in ways that may interact with graft selection [85, 89]. However, the identification of reliable preoperative biomarkers capable of predicting which patients are most likely to benefit from a given graft category remains at an early stage.
Serum biomarkers of bone turnover, including osteocalcin, procollagen type I N-terminal propeptide, and collagen degradation products, have been investigated as potential indicators of bone healing capacity and fracture nonunion risk [159, 160]. More recently, circulating collagen X fragments have been explored as quantitative markers of endochondral ossification activity during fracture repair, with preliminary evidence suggesting that such markers may correlate with healing trajectory [161]. The molecular profiling of fracture nonunion tissue has identified distinct biological signatures that distinguish successful from compromised repair environments, raising the possibility that tissue-level or circulating biomarkers could eventually inform graft selection decisions [162]. Despite these developments, no validated biomarker panel currently exists for guiding bone graft selection in clinical practice. The translation of candidate biomarkers from research settings to clinical decision support will require prospective validation studies, standardization of analytical platforms, and integration with clinical outcome data across diverse graft types and surgical contexts.
7.4 Longitudinal Registry Data and Real-World Evidence
The limitations of conventional clinical trial data for bone graft evaluation have prompted growing interest in registry-based approaches and real-world evidence as complementary sources of comparative effectiveness information [158]. Clinical registries have the capacity to capture longitudinal outcomes across large, heterogeneous patient populations under routine surgical conditions, providing data that may better reflect the performance of graft products in the full spectrum of clinical practice rather than in the controlled environments of efficacy trials. Registry infrastructure for orthopedic devices and biologics has expanded in recent years including the American Academy of Orthopaedic Surgeons (AAOS) American Joint Replacement Registry, the Quality Outcomes Database (QOD) maintained by the AANS/CNS for spine procedures, and international efforts such as the National Joint Registry of England, Wales, Northern Ireland and the Isle of Man,, though the incorporation of graft- specific outcome data into existing spine and trauma registries remains incomplete [103].
The development of product-specific outcome registries, in which graft type, manufacturer, formulation, and handling conditions are systematically recorded alongside patient outcomes, would represent a meaningful advance in the capacity to generate comparative evidence. Such registries could facilitate post-market surveillance, enable identification of product-level performance differences that are not apparent in aggregated class-level data, and provide the longitudinal follow- up necessary to detect differences in revision rates, fusion durability, and patient-reported outcomes that may not emerge within the follow-up windows of conventional studies. International efforts to establish coordinated registry networks for orthopedic interventions offer a potential framework, though the inclusion of detailed biologic graft data within these systems is not yet standard practice.
7.5 Emerging Technologies and Future Graft Strategies
Beyond improvements in evaluation methodology, the broader landscape of bone graft technology continues to evolve in ways that may reshape the comparative framework over time. Advances in three-dimensional bioprinting have introduced the possibility of patient-specific scaffold architectures with precisely controlled porosity, geometry, and compositional gradients, potentially enabling graft constructs that are tailored to specific defect characteristics and host conditions [107, 108, 112, 113, 121]. Gene-enhanced strategies, including regional gene therapy approaches using viral or non-viral delivery of osteogenic transgenes such as BMP-2, have demonstrated efficacy in preclinical models and are beginning to enter early-phase clinical investigation [163]. Synthetic bone graft materials are also evolving through the addition of bioactive components and the design of increasingly complex architectures intended to better regulate the local regenerative environment. Additives such as trace ions, polymer carriers, and composite ceramic phases may alter resorption kinetics, surface reactivity, and cell–material interactions, while engineered pore networks and micro- or nanoscale surface features can improve osteoconduction, vascular ingrowth, and mechanical integration [112]. These developments suggest that future comparative frameworks will need to account not only for biologic activity derived from donor tissue or viable cells, but also for the capacity of synthetic materials to achieve functional performance through controlled chemistry and structural design.
These emerging technologies are not yet positioned to displace established DBM or CBA platforms in current clinical practice, and some of their regulatory and commercialization pathways remain incompletely defined [109, 110, 111]. However, they highlight the dynamic nature of the field and underscore the importance of developing evaluation frameworks that are sufficiently flexible to accommodate new graft strategies as they mature, thus providing clinicians with options for the best graft for their patient(s). The comparative analysis presented in this review is intended not as a fixed ranking of existing products but as a methodological template for the structured, multi-domain evaluation of bone graft technologies, as the evidence base continues to expand [117, 125, 127].
8. Conclusion
The comparative analysis presented in this review underscores the fundamental challenge of rendering categorical judgments regarding the relative merits of demineralized bone matrix (DBM) and cellular bone allografts (CBAs) across diverse clinical applications. Both graft categories offer important biologic advantages and remain clinically relevant options within contemporary bone grafting strategies. DBM provides a widely used and biologically active matrix that combines osteoinductive potential with osteoconductive scaffold support, while CBAs are designed to build upon this framework through the addition of viable cellular components intended to enhance osteogenic contribution. Although these graft categories differ meaningfully in biologic rationale, the currently available clinical evidence does not establish consistent superiority of one category over the other across the range of surgical indications in which both are utilized. Rather, the scientific literature, including preclinical work, retrospective case studies, case series, and single-arm clinical investigations, supports clinical merit for both categories, while also indicating that further high-quality comparative evaluation is needed.
The clinical evidence supporting both graft categories is characterized by heterogeneity in study design, substantial reliance on retrospective and single-arm methodologies, variability in endpoint definitions and imaging-based fusion assessment criteria, and limited representation of rigorously controlled head-to-head comparisons. The evidence base for DBM benefits from greater depth and longer accrual, including meta- analytic synthesis, while CBA evidence, though growing, remains concentrated in single-arm prospective designs that do not yet permit the same level of confidence in cross-study generalization. Manufacturing and regulatory heterogeneity within each graft class further complicates interpretation, as products marketed under the same general category may differ in processing methods, carrier composition, cellular specifications, potency criteria, and regulatory pathway. These factors collectively limit the generalizability of product-specific results to broader graft categories and argue against reliance on class-level assumptions in clinical decision-making.
Graft selection in current clinical practice is therefore most appropriately guided by an integrated assessment that considers indication-specific clinical evidence, patient-level host biology and risk factors, regulatory transparency, manufacturing consistency, handling characteristics, and economic sustainability rather than categorical graft class preference alone. In this context, DBM remains attractive for its long history of clinical use, versatile handling formats, and established role as an extender or biologically active scaffold, whereas CBAs may offer value in settings where the addition of viable cellular elements is considered desirable within the overall graft construct. At present, however, graft selection is influenced at least as much by regulatory access, institutional formulary decisions, and perceived biological advantage as by definitive comparative evidence. The development of standardized potency assays, comparative clinical trial designs, biomarker-guided selection strategies, and product-specific outcome registries will be essential for advancing the evidence base toward the resolution necessary to support more precise, individualized graft selection. Until such data are available, the field would benefit from greater transparency regarding the evidentiary basis supporting individual products and a clearer distinction between what current graft technologies are designed to achieve and what they have been shown to deliver under controlled conditions.
Author Contributions: Both authors contributed equally to this project. All authors have read and agreed to the published version of the manuscript.
Acknowledgments: The authorship group would like to formally acknowledge Mr. Andrew Jones for his diligence in creating the product comparison table.
Funding: This research received no external funding.
Informed Consent Statement: Informed consent was not required for this study in accordance with applicable institutional and federal regulations, as no identifiable human subjects or protected health information were involved.
Conflicts of Interest: The authors have the following relevant disclosures: Bryce Jewett is a paid consultant of Staysis Medical. Aaron J. Tabor is employed by Staysis Medical and MedTech Incubator. Both companies offer services or bone products.
References
- Urist MR. (1965). Bone: formation by autoinduction. Science.
- Reddi AH, Huggins C. (1972). Biochemical sequences in the transformation of normal fibroblasts in adolescent rats. Proc Natl Acad Sci USA.
- Wozney JM et al.. (1988). Novel regulators of bone formation: molecular clones and activities. Science.
- Albrektsson T, Johansson C. (2001). Osteoinduction, osteoconduction and osseointegration. Eur Spine J.
- Khan SN et al.. (2005). The biology of bone grafting. J Am Acad Orthop Surg.
- Giannoudis PV et al.. (2005). Bone substitutes: an update. Injury.
- Salhotra A et al.. (2020). Mechanisms of bone development and repair. Nat Rev Mol Cell Biol.
- Chen D et al.. (2004). Bone morphogenetic proteins. Growth Factors.
- Carreira AC et al.. (2014). Bone morphogenetic proteins: facts, challenges, and future perspectives. J Dent Res.
- Katagiri T, Watabe T. (2016). Bone morphogenetic proteins. Cold Spring Harb Perspect Biol.
- Carano RAD, Filvaroff EH. (2003). Angiogenesis and bone repair. Drug Discov Today.
- Claes L et al.. (2012). Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol.
- Marsell R, Einhorn TA. (2011). The biology of fracture healing. Injury.
- Oryan A et al.. (2014). Bone morphogenetic proteins: a powerful osteoinductive compound with non-negligible side effects and limitations. Biofactors.
- Wang W, Yeung KWK. (2017). Bone grafts and biomaterials substitutes for bone defect repair: a review. Bioact Mater.
- Gruskin E et al.. (2012). Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev.
- Drosos GI et al.. (2006). Factors affecting fracture healing after intramedullary nailing of the tibial diaphysis for closed and grade I open fractures. J Bone Joint Surg Br.
- Zhang H et al.. (2019). Demineralized bone matrix carriers and their clinical applications: an overview. Orthop Surg.
- Bae HW et al.. (2006). Osteoinductive ability of human allograft formulations. Journal of Periodontology.
- Bae H et al.. (2010). Variability across ten lots of a commercially available demineralized bone matrix product. J Bone Joint Surg Am.
- Peterson B et al.. (2004). Osteoinductivity of commercially available demineralized bone matrix: preparations in a spine fusion model. J Bone Joint Surg Am.
- Katz JM et al.. (2009). Demineralized bone matrix as an osteoinductive biomaterial and in vitro predictors of its biological potential. J Biomed Mater Res B.
- Pietrzak WS, Woodell-May JE, McDonald N. (2006). Assay of bone morphogenetic protein-2, -4, and -7 in human demineralized bone matrix. Journal of Craniofacial Surgery.
- Holt DJ, Grainger DW. (2012). Demineralized bone matrix as a vehicle for delivering endogenous and exogenous therapeutics in bone repair. Adv Drug Deliv Rev.
- Eleswarapu A et al.. (2021). Efficacy, Cost, and Complications of Demineralized Bone Matrix in Instrumented Lumbar Fusion: Comparison With rhBMP-2. Global Spine Journal.
- Fu TS et al.. (2016). Fusion rate of DBM vs autogenous iliac bone graft for long multisegment posterolateral fusion. BMC Musculoskeletal Disorders.
- Nam HS, Yi JS. (2016). Bone union rate after instrumented posterolateral lumbar fusion using DBM vs Hydoxyapetite. Asian Spine J.
- Chang DG et al.. (2021). Surgical outcomes of two DBM putties/local autograft composites in instrumented posterolateral lumbar fusion. BMC Musculoskelet Disord.
- Han S et al.. (2020). Meta-analysis of DBM + autograft vs autograft alone in lumbar fusion. J Korean Neurosurg Soc.
- Heard J et al.. (2023). Impact of DBM characteristics on pseudarthrosis after posterolateral fusion. J Craniovertebral Junction Spine.
- Buser Z et al.. (2018). Allograft vs demineralized bone matrix in instrumented and non instrumented lumbar fusion: systematic review. Global Spine J.
- Kim SJ et al. (2017). Polyetheretherketone Cage with Demineralized Bone Matrix Can Replace Iliac Crest Autografts for Anterior Cervical Discectomy and Fusion in Subaxial Cervical Spine Injuries. J Korean Neurosurg Soc.
- Ajiboye RM et al.. (2016). Outcomes of DBM enriched with concentrated bone marrow aspirate in lumbar fusion. Int J Spine Surg.
- Staysis Medical. (2025). Staysis DBM Putty and DBM Crunch: product documentation and specifications. StaysisMedical, Scottsdale, AZ.
- Chin KR et al.. (2017). Soft tissue swelling incidence using DBM in outpatient ACDF. World J Orthop.
- Aghdasi B et al.. (2013). A review of demineralized bone matrices for spinal fusion: the evidence for efficacy. Surgeon.
- Cammisa FP Jr et al.. (2004). Two-year fusion rate equivalency between Grafton DBM gel and autograft in posterolateral spine fusion: a prospective controlled trial employing a side-by-side comparison in the same patient. Spine.
- Tilkeridis K et al.. (2012). Use of demineralized bone matrix in spinal fusion. World J Orthop.
- Zadegan SA et al.. (2017). Demineralized bone matrix (DBM) in anterior cervical discectomy and fusion: systematic review. Eur Spine J.
- Lee TC et al.. (2005). The effect of acute smoking on spinal fusion: an experimental study among rabbits. J Trauma
- Pieske O et al.. (2009). Autologous bone graft versus demineralized bone matrix in internal fixation of ununited long bones. J Trauma Manag Outcomes.
- Drosos GI et al.. (2020). The choice between allograft or demineralized bone matrix is not unambiguous in trauma surgery. Injury.
- Grabowski G, Cornett CA. (2013). Bone graft and bone graft substitutes in spine surgery: current concepts and controversies. J Am Acad Orthop Surg.
- Bae HW et al.. (2006). Intervariability and intravariability of bone morphogenetic proteins in commercially available demineralized bone matrix products. Spine.
- Russell JL. (2000). Grafton demineralized bone matrix: performance consistency, utility, and value. Tissue Eng.
- Russell N et al.. (2020). In-vivo performance of seven commercially available demineralized bone matrix fiber and putty products in a rat posterolateral fusion model. Frontiers in Surgery.
- Pittenger MF et al.. (1999). Multilineage potential of adult human mesenchymal stem cells. Science.
- Caplan AI. (1991). Mesenchymal stem cells. J Orthop Res.
- Dominici M et al.. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells: ISCT position statement. Cytotherapy.
- Caplan AI, Dennis JE. (2006). Mesenchymal stem cells as trophic mediators. J Cell Biochem.
- Gnecchi M et al.. (2016). Paracrine mechanisms of mesenchymal stem cells in tissue repair. Methods Mol Biol.
- Liang X et al.. (2014). Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant.
- Marquez-Curtis LA et al.. (2015). Mesenchymal stromal cells derived from various tissues: biological, clinical, and cryopreservation aspects. Cryobiology.
- Moll G et al.. (2014). Do cryopreserved mesenchymal stromal cells display impaired immunomodulatory and therapeutic properties?. Stem Cells.
- Hernigou P et al.. (2005). Percutaneous autologous bone- marrow grafting for nonunions: influence of the number and concentration of progenitor cells. J Bone Joint Surg Am.
- Bruder SP et al.. (1998). Mesenchymal stem cells in osteobiology and applied bone regeneration. Clin Orthop Relat Res.
- Lansford TJ et al.. (2024). High lumbar spinal fusion rates using cellular bone allograft irrespective of surgical risk factors. Int J Spine Surg.
- Russo GS et al.. (2024). Impact of surgical risk factors for non- union on lumbar spinal fusion rates using cellular bone allograft. BMC Musculoskelet Disord.
- Elgafy H et al.. (2021). Lumbar spine fusion using V-CBA (lineage-committed bone-forming cells). BMC Musculoskelet Disord.
- Goldman SM et al.. (2024). Allograft cellular bone matrix for anterior cervical discectomy and fusion. J Spine Surg.
- Bergin PF et al.. (2021). Pseudarthrosis rate following ACDF using Osteocel. Clin Spine Surg.
- Gibson S et al.. (2002). Allograft versus autograft in instrumented posterolateral lumbar spinal fusion. Spine.
- Hsieh PC et al.. (2019). Allogenic stem cells in spinal fusion: systematic review. Global Spine J.
- Kim PD et al.. (2023). 12-Month clinical and radiographic outcomes of ViBone viable bone matrix in patients undergoing cervical and lumbar spinal fusion surgery. J Orthop Surg Res.
- Tohmeh AG et al.. (2012) Allograft cellular bone matrix in extreme lateral interbody fusion: preliminary radiographic and clinical outcomes. ScientificWorldJournal.
- Kerr EJ III et al.. (2011). The use of osteo-conductive stem-cells allograft in lumbar interbody fusion procedures: an alternative to rhBMP. J Surg Orthop Adv.
- Saitta B et al.. (2021) Cellular bone matrix in multilevel ACDF: A prospective evaluation of 1-year fusion rates and patient reported outcomes. The Spine Journal.
- Hollawell SM. (2012). Allograft cellular bone matrix as an alternative to autograft in hindfoot and ankle fusion procedures. J Foot Ankle Surg.
- Divi SN et al.. (2021). Investigating the efficacy of allograft cellular bone matrix for spinal fusion: systematic review of the literature. Neurosurg Focus.
- Jones CP et al.. (2015). Prospective, multicenter evaluation of allogeneic bone matrix containing viable osteogenic cells in foot and ankle arthrodesis procedures. Foot Ankle Int.
- Eastlack RK et al.. (2014). Osteocel Plus cellular allograft in anterior cervical discectomy and fusion: evaluation of clinical and radiographic outcomes from a prospective multicenter study. Spine.
- Vanichkachorn J et al.. (2016). A prospective clinical and radiographic 12-month outcome study of patients undergoing single-level anterior cervical discectomy and fusion for symptomatic cervical degenerative disc disease utilizing a novel viable allogeneic, cancellous, bone matrix (Trinity Evolution) with a comparison to historical controls. Eur Spine J.
- Lin C et al.. (2020). Comparing cellular bone matrices for posterolateral spinal fusion in a rat model. JOR Spine.
- Dhaliwal J et al.. (2025). A prospective evaluation of cellular bone matrix for posterolateral lumbar fusion. Clinical Neurology and Neurosurgery.
- Singh, Samradhi et al. (2023) From Cells to Environment: Exploring the Interplay between Factors Shaping Bone Health and Disease. Medicina (Kaunas, Lithuania)
- Colnot C. (2009). Skeletal cell fate decisions within periosteum and bone marrow during bone regeneration. J Bone Miner Res.
- Lambrechts MJ et al.. (2024). Cellular bone matrix in spine surgery: are they worth the risk? A systematic review. Global Spine J.
- Bhamb N et al.. (2019). Comparative efficacy of commonly available human bone graft substitutes as tested for posterolateral fusion in an athymic rat model. Int J Spine Surg.
- Tuchman A et al.. (2016). Iliac crest bone graft versus local autograft or allograft for lumbar spinal fusion: a systematic review. Global Spine J.
- Kadam A et al.. (2016). Bone substitutes and expanders in spine surgery: a review of their fusion efficacies. Int J Spine Surg.
- Kaiser MG et al.. (2014). Bone graft materials in lumbar spine fusion: recommendations from the Joint Section on Disorders of the Spine and Peripheral Nerves of the AANS/CNS. J Neurosurg Spine.
- Stephan SR et al.. (2021). Stem cells and spinal fusion. Int J Spine Surg.
- Li S et al.. (2022). A systematic review and meta-analysis of fusion rate enhancements and bone graft options for spine surgery. Sci Rep.
- Dimitriou R et al.. (2011). Bone regeneration: current concepts and future directions. BMC Med.
- Giannoudis PV et al.. (2007). Fracture healing in osteoporotic fractures: is it really different? A basic science perspective. Injury.
- Nauth A et al.. (2010). Growth factors: beyond bone morphogenetic proteins. J Orthop Trauma.
- Sen MK, Miclau T. (2007). Autologous iliac crest bone graft: should it still be the gold standard for treating nonunions?. Injury.
- Calori GM et al.. (2011). The use of bone-graft substitutes in large bone defects: any specific needs?. Injury.
- Berman D et al.. (2017). The effect of smoking on spinal fusion. Int J Spine Surg.
- Arnold PM, Harrop JS, Mariscal G, Sasso RC, Steinmetz MP, Chaput CD, Jacobs WB, Witiw CD, O’Toole JE. (2025). Adverse impact of smoking on spine fusion and patient- reported outcomes: a systematic review and meta-analysis. Global Spine Journal.
- Steinmetz MP, Arnold PM, Harrop JS, Mariscal G, Sasso RC, Chaput CD, Jacobs WB, Witiw CD, O’Toole JE. (2025). Adverse impact of diabetes on spine fusion and patient- reported outcomes: a systematic review and meta-analysis. Spine.
- Boonsirikamchai W et al.. (2024). Pseudarthrosis risk factors in lumbar fusion: a systematic review and meta-analysis. BMC Musculoskelet Disord.
- Ackerman SJ et al.. (2002). Economic evaluation of bone morphogenetic protein versus autogenous iliac crest bone graft in single-level anterior lumbar fusion. Spine.
- Carreon LY et al.. (2013). Cost-effectiveness of single-level anterior cervical discectomy and fusion five years after surgery. Spine.
- Glassman SD et al.. (2012). Cost effectiveness analysis of graft options in spinal fusion surgery using a Markov model. The Spine Journal.
- Gallagher KK et al.. (2023). Osteobiologics and value-based care: challenges and opportunities. Int J Spine Surg.
- Amini AR et al.. (2012). Bone Tissue Engineering: Recent Advances and Challenges. Crit Rev Biomed Eng.
- Sipp D et al.. (2017). Marketing of unproven stem cell-based interventions: a call to action. Sci Transl Med.
- Viswanathan S et al. (2019) Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT ) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy.
- Scarfe L et al.. (2017). Preclinical imaging methods for assessing the safety and efficacy of regenerative medicine therapies. NPJ Regen Med.
- FDA. (2017). Regulatory considerations for human cells, tissues, and cellular and tissue-based products: minimal manipulation and homologous use. FDA Guidance Document.
- McGowan KB, Stiegman G. (2013). Regulatory challenges for cartilage repair technologies. Cartilage.
- ISASS Policy Statement. (2019). Recommendations and coverage criteria for bone graft substitutes used in spinal surgery. Int J Spine Surg.
- Boden SD. (2002). Overview of the biology of lumbar spine fusion and principles for selecting a bone graft substitute. Spine.
- Gillman CE, Jayasuriya AC. (2021). FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Materials (Basel).
- Loi F et al.. (2016). Inflammation, fracture and bone repair. Bone.
- Perez JR et al.. (2018). Tissue engineering and cell-based therapies for fractures and bone defects. Front Bioeng Biotechnol.
- Stahl A, Yang YP. (2021). Regenerative approaches for the treatment of large bone defects. Tissue Eng Part B Rev.
- Gao C et al.. (2014). Current progress in bioactive ceramic scaffolds for bone repair and regeneration. Int J Mol Sci.
- Roseti L et al.. (2017). Scaffolds for bone tissue engineering: state of the art and new perspectives. Mater Sci Eng C.
- Vidal L et al.. (2020). Reconstruction of large skeletal defects: current clinical therapeutic strategies and future directions using 3D printing. Front Bioeng Biotechnol.
- Bose S et al.. (2012). Recent advances in bone tissue engineering scaffolds. Trends Biotechnol.
- Tatara AM, Mikos AG. (2016). Tissue engineering in orthopaedics. J Bone Joint Surg Am.
- Arrington ED et al.. (1996). Complications of iliac crest bone graft harvesting. Clin Orthop Relat Res.
- Younger EM, Chapman MW. (1989). Morbidity at bone graft donor sites. J Orthop Trauma.
- Van der Stok J et al.. (2017). The available evidence on demineralised bone matrix in trauma and orthopaedic surgery. Bone Joint Res.
- Mediouni M et al.. (2023). Current options and future perspectives on bone graft and biomaterials substitutes for bone repair. Appl Sci.
- Biddau F et al.. (2024). Bone graft substitutes in anterior lumbar interbody fusion: a systematic review. Global Spine J.
- Yang S et al.. (2024). Risk factors affecting spinal fusion: a meta-analysis. PLoS One.
- Olivares-Navarrete R et al.. (2012). Osteogenic differentiation of stem cells alters vitamin D receptor expression. Stem Cells Dev.
- Koons GL et al.. (2020). Materials design for bone tissue engineering. Nat Rev Mater.
- De Long WG Jr et al.. (2007). Bone grafts and bone graft substitutes in orthopaedic trauma surgery: a critical analysis. J Bone Joint Surg Am.
- Boyce T et al.. (1999). Allograft bone: the influence of processing on safety and performance. Orthop Clin North Am.
- Greenwald AS et al.. (2001). Bone-graft substitutes: facts, fictions, and applications. J Bone Joint Surg Am.
- Campana V et al.. (2014). Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med.
- Fillingham Y, Jacobs J. (2016). Bone grafts and their substitutes. Bone Joint J.
- Fernandez de Grado G et al.. (2018). Bone substitutes: a review of their characteristics, clinical use, and perspectives for large bone defects management. J Tissue Eng.
- Betz RR. (2002). Limitations of autograft and allograft: new synthetic surfaces. Orthopedics.
- Finkemeier CG. (2002). Bone-grafting and bone-graft substitutes. J Bone Joint Surg Am.
- Dallari D et al.. (2007). Enhanced tibial osteotomy healing with use of bone grafts supplemented with platelet gel or platelet gel and bone marrow stromal cells. J Bone Joint Surg Am.
- Skovrlj B et al.. (2014). Cellular bone matrices: viable stem cell-containing bone graft substitutes. Spine J.
- Hsu WK et al.. (2008). Stem cells from human fat as cellular delivery vehicles in an athymic rat posterolateral spine fusion model. J Bone Joint Surg Am.
- Gupta MC et al.. (2007). Efficacy of mesenchymal stem cell enriched grafts in an ovine posterolateral lumbar spine model. Spine.
- Muschler GF et al.. (2010). The design and use of animal models for translational research in bone tissue engineering and regenerative medicine. Tissue Eng Part B Rev.
- Deyo RA et al.. (2010). Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA.
- Fischer CR et al.. (2013). A systematic review of comparative studies on bone graft alternatives for common spine fusion procedures. European Spine Journal.
- RTI Surgical. (2014). Map3 Cellular Allogeneic Bone Graft: product characterization and clinical application summary. RTI Surgical product documentation.
- Lubelski D, Abdullah KG, Benzel EC, Mroz TE. (2012). The utility of allograft mesenchymal stem cells for spine fusion: a literature review. Global Spine Journal.
- Zakko P et al.. (2023). Anterior cervical discectomy and fusions supplemented with cellular or noncellular allografts have similar radiographic fusion and clinical outcomes. Clin Spine Surg.
- Tuchman A, Brodke DS, Youssef JA, Meisel HJ, Dettori JR, Park JB, Yoon ST, Wang JC. (2017). Autograft versus allograft for cervical spinal fusion: a systematic review. Global Spine Journal.
- Keating JF, McQueen MM. (2001). Substitutes for autologous bone graft in orthopaedic trauma. Journal of Bone and Joint Surgery (British).
- Stryker. (2019). Bio4 Bone Matrix: product overview and clinical summary. Stryker product documentation.
- Gianulis et al.. (2023). Characterization of an advanced viable bone allograft with preserved native bone-forming cells. Cell and Tissue Banking.
- Zhang J et al.. (2025). Research progress of bone grafting: a comprehensive review. International Journal of Nanomedicine.
- Buser Z et al.. (2016). Synthetic bone graft versus autograft or allograft for spinal fusion: a systematic review. Journal of Neurosurgery: Spine.
- Ramirez S, Coughlin D, Clough B, Sederberg J, Antin SP. (2017). Four Food and Drug Administration draft guidance documents and the REGROW Act: a litmus test for future changes in HCT/P regulatory policy. Tissue Engineering Part B: Reviews.
- Jeyaraman M et al.. (2024). Evidence-based orthobiologic practice: current evidence review and future directions. World J Orthop.
- Thaci et al.. (2021). Cost-effectiveness of peptide enhanced bone graft i-Factor versus use of local autologous bone in ACDF surgery. Clinicoecon Outcomes Res.
- Cowell et al.. (2023). Cost-effectiveness of decellularised bone allograft compared with fresh-frozen bone allograft for acetabular impaction bone grafting. BMJ Open.
- Liu Y, Park CW, Pholprajug P, Suvithayasiri S, Kim JH, Lee C, Kim E, Kim JS. (2025). Efficacy of allograft versus bioactive glass-ceramic cage in anterior cervical discectomy and fusion: a randomized controlled study. SAGE Open Medicine.
- Chu CR et al. (2019). Optimizing clinical use of biologics in orthopaedic surgery: consensus recommendations from the 2018 AAOS/NIH U-13 conference. Journal of the American Academy of Orthopaedic Surgeons.
- Han B, Yang Z, Nimni ME. (2008). Effects of gamma irradiation on osteoinduction associated with demineralized bone matrix. Journal of Orthopaedic Research.
- Nguyen H, Morgan DAF, Forwood MR. (2006). Sterilization of allograft bone: is 25 kGy the gold standard for gamma irradiation?. Cell and Tissue Banking.
- Bahsoun S, Coopman K, Akam EC. (2019). The impact of cryopreservation on bone marrow-derived mesenchymal stem cells: a systematic review. Journal of Translational Medicine.
- Galipeau J, Sensébé L. (2018). Mesenchymal stromal cells: clinical challenges and therapeutic opportunities. Cell Stem Cell.
- Chinnadurai R, Rajan D, Qayed M, Arafat D, Garcia M, Liu Y, Kugathasan S, Anderson LJ, Gibson G, Galipeau J. (2018). Potency analysis of mesenchymal stromal cells using a combinatorial assay matrix approach. Cell Reports.
- Hubbell PJ, Roth B, Block JE. (2023). Comparative evaluation of mineralized bone allografts for spinal fusion surgery. Journal of Functional Biomaterials.
- Magaway CL, Bhatt M, Murray IR. (2022). Patient registries in orthopedics and orthobiologic procedures: a narrative review. BMC Musculoskeletal Disorders.
- Perut F, Roncuzzi L, Gómez-Barrena E, Baldini N. (2024). Association between bone turnover markers and fracture healing in long bone non-union: a systematic review. Journal of Clinical Medicine.
- Chitwood JR, Chakraborty N, Hammamieh R, Moriarity KA, Swiontkowski MF, Christensen HC. (2021). Predicting fracture healing with blood biomarkers: the potential to assess patient risk of fracture nonunion. Biomarkers.
- Working ZM, Morris ER, Chang JC, Warman ML, Herndon DN, Bhatt S. (2021). A quantitative serum biomarker of circulating collagen X effectively correlates with endochondral fracture healing. Journal of Orthopaedic Research.
- Panteli M, Vun JSH, Pountos I, Howard JA, Jones E, Giannoudis PV. (2022). Biological and molecular profile of fracture non- union tissue: a systematic review and an update on current insights. Journal of Cellular and Molecular Medicine.
- de la Vega RE, Atasoy-Zeybek A, Panos JA, van Griensven M, Evans CH, Balmayor ER. (2021). Gene therapy for bone healing: lessons learned and new approaches. Translational Research.
- Rajaee SS, Bae HW, Kanim LEA, Delamarter RB. (2012). Spinal fusion in the United States: analysis of trends from 1998 to 2008. Spine.
- Roberts TT, Rosenbaum AJ. (2012). Bone grafts, bone substitutes and orthobiologics: the bridge between basic science and clinical advancements in fracture healing. Organogenesis.
- Vaccaro AR et al.. (2004). A pilot study evaluating the safety and efficacy of OP-1 putty (rhBMP-7) as a replacement for iliac crest autograft in posterolateral lumbar arthrodesis for degenerative spondylolisthesis. Spine.
- Sayeed SA et al.. (2024). Radiographic fusion outcomes for Trinity CBA vs local bone in posterolateral lumbar fusion. JAAOS Glob Res Rev.
- Peppers TA et al.. (2017). Prospective clinical and radiographic evaluation of an allogeneic bone matrix containing stem cells (Trinity Evolution) in patients undergoing two-level ACDF. J Orthop Surg Res.
- McAnany SJ et al.. (2016). Mesenchymal stem cell allograft as a fusion adjunct in one- and two-level anterior cervical discectomy and fusion: a matched cohort analysis. Spine J.
- Dimar JR II et al. (2006). Clinical outcomes and fusion success at 2 years of single-level instrumented posterolateral fusions with recombinant human bone morphogenetic protein-2/compression resistant matrix versus iliac crest bone graft. Spine.
- Vaccaro AR et al.. (2008). Comparison of OP-1 putty (rhBMP- 7) to iliac crest autograft for posterolateral lumbar arthrodesis: a minimum 2-year follow-up pilot study. Spine.
- Chau AMT, Mobbs RJ. (2009). Bone graft substitutes in anterior cervical discectomy and fusion. Eur Spine J.
- Miyazaki M et al.. (2008). Comparison of human mesenchymal stem cells derived from adipose tissue and bone marrow for ex vivo gene therapy in rat spinal fusion model. Spine.
- Bydon M et al.. (2014). The current role of steroids in acute spinal cord injury. WorldNeurosurg.
- Sasso RC et al.. (2004). A prospective, randomized controlled clinical trial of anterior lumbar interbody fusion using a titanium cylindrical threaded fusion device. Spine.
- Boden SD et al.. (1999). Posterolateral lumbar intertransverse process spine arthrodesis with recombinant human bone morphogenetic protein 2/hydroxyapatite-tricalcium phosphate after laminectomy in the nonhuman primate. Spine.
- Burkus JK et al.. (2002). Anterior lumbar interbody fusion using rhBMP-2 with tapered interbody cages. J Spinal Disord Tech.
- Simmonds MC et al.. (2013). Safety and effectiveness of recombinant human bone morphogenetic protein-2 for spinal fusion: a meta-analysis of individual-participant data. Ann Intern Med.
- Fu R et al.. (2013). Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med.
- Carragee EJ et al.. (2011). A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: emerging safety concerns and lessons learned. Spine J.
- Zhang Y et al.. (2016). Lentivirus-TAZ Administration Alleviates Osteoporotic Phenotypes in the Femoral Neck of Ovariectomized Rats. Cell Physiol Biochem.
- Boden SD et al.. (2000). Use of recombinant human bone morphogenetic protein-2 to achieve posterolateral lumbar spine fusion in humans: a prospective, randomized clinical pilot trial. Spine.
- Malham GM, Scheuber T, Williams RP, Ellis TJ, Egeler RM, Ellis SJ. (2022). Recombinant human bone morphogenetic protein-2 in spine surgery: recommendations for use and alternative bone substitutes: a narrative review. Journal of Spine Surgery.
- Wang JC et al. (2007). A comparison of commercially available demineralized bone matrix for spinal fusion. Eur Spine J.
- Ghobrial GM, Anderson KD, Maulucci CM, Vachhani JA, Darden BV, Arnold PM. (2023). Characterization of an advanced viable bone allograft: cellular composition, osteogenic potential, and clinical evidence. Cell and Tissue Banking.
- Musante DB, Firtha ME, Atkinson BL, Heffner R, Koenig AL, Rofail SS. (2016). Clinical evaluation of an allogeneic bone matrix containing viable osteogenic cells in patients undergoing posterolateral lumbar arthrodesis. J Spine Surg.undergoing one- and two-level posterolateral lumbar arthrodesis with decompression. J Spine Surg.
- Buser Z, Brodke DS, Youssef JA, Meisel HJ, Park JB, Yoon ST, Wang JC. (2020). Comparing cellular bone matrices for posterolateral spinal fusion in a rat model. J Biomater Appl.
- U.S. Food and Drug Administration. (2008). 510(k) Summary: Progenix Plus DBM (K081950). FDA.
- U.S. Food and Drug Administration. (2013). 510(k) Summary: Magnifuse Bone Graft (K123691). FDA.
- U.S. Food and Drug Administration. (2007). 510(k) Summary: AlloFuse DBM (K071849). FDA.
- U.S. Food and Drug Administration. (2018). 510(k) Summary: InterGro DBM Plus (K171858). FDA.
- Zimmer Biomet Spine. (2015). A clinical study of the Cellentra VCBM/MaxAn anterior cervical plate system for anterior cervical discectomy and fusion. ClinicalTrials.gov (NCT02182843).
- SeaSpine. (2012). A prospective, multi-center study evaluating Accell Evo3 in posterolateral spine fusion. ClinicalTrials.gov (NCT01714804).
 Alcrut
Alcrut